Correct option is A
Explanation-
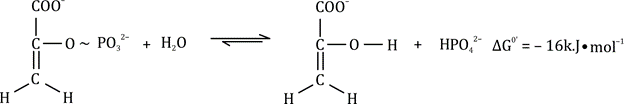
Reaction catalyzed by pyruvate kinase:
PEP + ADP → Pyruvate + ATP
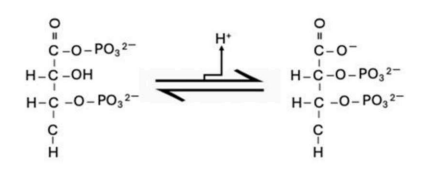
The enol form of pyruvate tautomerizes to its keto form.
Free energy changes:
Hydrolysis of PEP: ΔG°′ = -61.9 kJ/mol
ATP synthesis from ADP + Pi: ΔG°′ = +30.5 kJ/mol
This shows that the highly exergonic hydrolysis of PEP drives the formation of ATP.
The conversion of PEP to pyruvate involves keto-enol tautomerism, where enol pyruvate spontaneously tautomerizes to the more stable keto form. This tautomerization is coupled with ATP formation, and it releases enough energy to make the reaction overall favorable.
Option A - "Through coupling with keto-enol tautomerism where the enol form of pyruvate is converted to the keto form."
This directly describes the favorable tautomerization that drives ATP synthesis. This tautomerism releases free energy, which contributes to the overall energy balance of ATP formation. This is exactly what happens in the pyruvate kinase-catalyzed step of glycolysis.
Incorrect options-
Option B - "Through condensation of Pi with ADP"
Describes the chemical process of ATP formation but doesn’t explain the source of the energy—i.e., the coupling with tautomerization.
Option C - "Through linking to proton motive force"
Applies to oxidative phosphorylation, not glycolysis.
Option D - "Through coupling with hydrolysis of PPi"
Not relevant to this step. PPi hydrolysis is involved in biosynthetic reactions like DNA/RNA synthesis, not in pyruvate kinase action.
So, the correct answer is Option A -Through coupling with keto-enol tautomerism where the enol form of pyruvate is converted to the keto form.