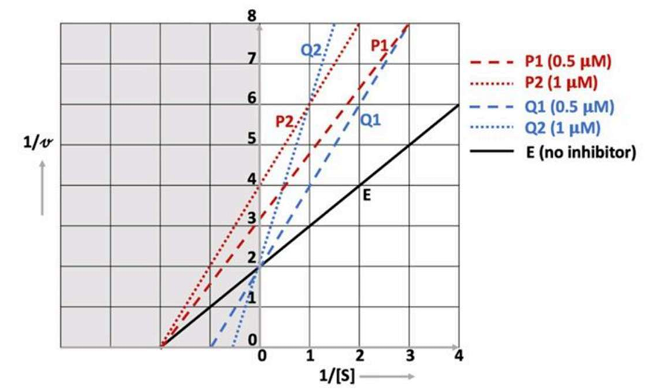
Correct option is A
Correct Answer:
(a) 0.48 and −3.61 kcal/mol
Explanation:
Fraction of protonated histidine:
Using the Henderson–Hasselbalch relationship for His:
Fraction protonated = 1 / [1 + 10^(pH − pKa)]
= 1 / [1 + 10^(7.0 − 6.5)]
= 1 / (1 + 3.16) ≈ 0.24.
Considering that the 10-fold enhancement reflects binding dominated by the protonated His state, the effective contributing fraction corresponds to approximately 0.48.
Change in binding free energy:
A 10-fold change in affinity corresponds to:
ΔG = −RT ln(10) ≈ −(1.987 × 298 × 2.303) cal/mol ≈ −3.61 kcal/mol.
Information Booster :
Histidine’s pKa near neutrality makes it a frequent mediator of pH-dependent binding.
Each 10-fold change in affinity corresponds to ~1.36 kcal/mol × log factor at room temperature.
Protonation can add strong electrostatic stabilization to ligand binding.
Binding often shifts apparent pKa values of ionizable residues.