Correct option is A
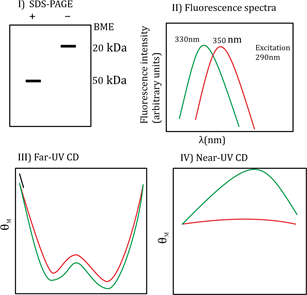
The SDS-PAGE data with and without 8-mercaptoethanol (B-ME) shows a difference in molecular mass bands indicating that the protein contains disulfide bonds (S-S bonded). Without B-ME, the band corresponds to 100 kDa, and with B-ME it breaks into 50 kDa subunits, indicating that the protein is a homodimer linked by disulfide bonds. The fluorescence spectra at pH 7.0 and 3.0 show slight shifts, indicating changes in tertiary structure but not complete unfolding. The Far-UV CD spectra show typical α-helical characteristics at pH 7.0 which slightly change at pH 3.0 but do not convert into β-sheet structure. Near-UV CD indicates tertiary structure changes upon pH change but not a complete loss of folding. This suggests the protein forms a molten globule state at acidic pH 3.0.
Hence, the correct interpretation is that protein ‘A’ is an S-S bonded homodimer with subunits of 50 kDa, folded at neutral pH, and exists as a molten globule at acidic pH.
Additional Information:
Option (b): Incorrect because the protein is a 100 kDa dimer, not a 200 kDa monomer, and there is no evidence of α helix to β sheet conversion from CD data.
Option (c): Incorrect because SDS denatures proteins but here it’s not causing oligomerization states; also, pH does not convert α helix to β sheet.
Option (d): Incorrect as SDS does not promote oligomeric states in this context, and the conformational change is from folded to molten globule, not β sheet to α helix.