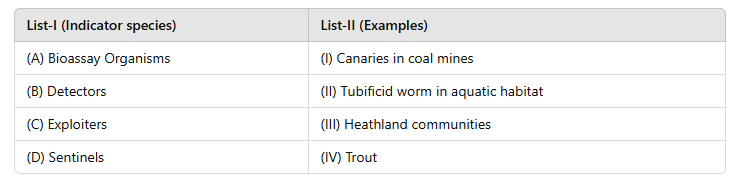
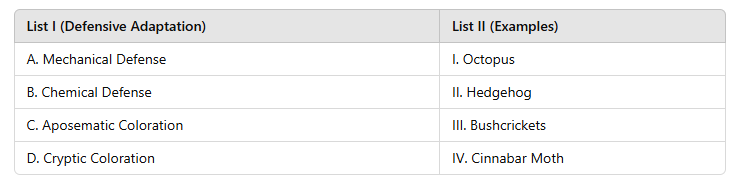
Correct option is B
In the process of biodiesel production, methanol is commonly used along with sodium hydroxide (NaOH) as a catalyst. The process is known as transesterification. Here's how it works:
1. Methanol reacts with the triglycerides in vegetable oil in the presence of sodium hydroxide to produce biodiesel (methyl esters) and glycerol as a by-product.
2. Sodium hydroxide acts as a catalyst, helping to break down the oil molecules and facilitating the chemical reaction.
This process ensures the conversion of vegetable oil into biodiesel, a renewable and eco-friendly fuel.
Additional Knowledge: 1. Methanol (Option 2):
· A simple alcohol used in transesterification.
· Mixed with NaOH or KOH to create a methoxide solution for the reaction.
· Key reason for use: affordable and effective.
2. Glycerol (Option 1):
· Not used in the reaction but is a by-product of biodiesel production.
· Has applications in pharmaceuticals, cosmetics, and food industries.
3. Ethanol (Option 3):
· Can also be used in biodiesel production (ethyl esters instead of methyl esters).
· Less commonly used due to cost and availability compared to methanol.
4. Xylene (Option 4):
· A hydrocarbon solvent, unrelated to biodiesel production.
· Commonly used in chemical industries for thinning paints and as a cleaning agent.





 English
English 10 Questions
10 Questions 12 Mins
12 Mins