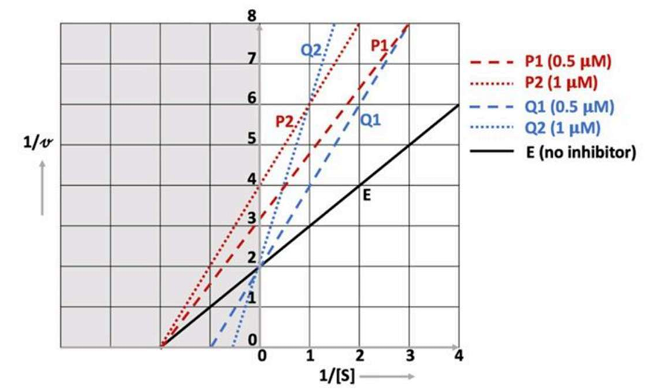
Correct option is A
The correct option is (a)
EXPLANATION-
Step 1: Understand pH and [H⁺] relationship
pH is defined as:
pH=−log[H+]
where [H⁺] is the concentration of hydrogen ions in moles per liter (M).
Using this formula, we calculate the initial and final hydrogen ion concentrations.
Step 2: Calculate initial [H⁺] concentration
Given initial pH = 7.65
[H+]initial=10−7.65
Using a calculator
[H+]initial=2.24×10−8 M
Step 3: Calculate final [H⁺] concentration
Given final pH = 6.87
[H+]final=10−6.87
Using a calculator
[H+]final=1.35×10−7 M
Step 4: Calculate the change in [H⁺]
Δ[H+]=[H+]final−[H+]initial =1.35×10−7−2.24×10−8=1.13×10−7M
This increase in hydrogen ion concentration corresponds to the number of moles of acetylcholine hydrolyzed.
Step 5: Calculate the number of moles of acetylcholine in 15 mL
Convert 15 mL to liters:
15 mL=0.015 L
Number of moles of [H⁺] (or acetylcholine hydrolyzed) in solution:
n=Δ[H+]×Volume (L)=1.13×10−7×0.015=1.695×10−9 mol
Step 6: Compare with options
The calculated value 1.695×10−9mol lies between
1.65×10−9 mol to 1.75×10−9 mol