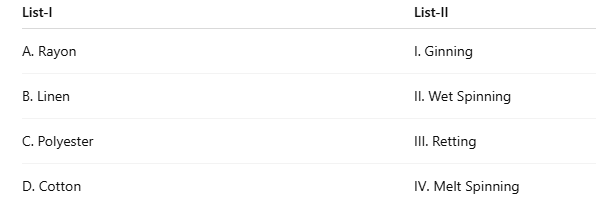
Correct option is C
The formula for
Normality (N) is given by:
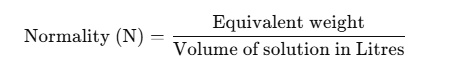
Normality is a measure of concentration that accounts for the equivalent weight of a substance in solution, which depends on the chemical reactions it can participate in (such as acid-base reactions or redox reactions). Normality is used for titrations and involves the volume of the solution in liters and the equivalent weight of the solute.
Information Booster: Normality (N) refers to the number of equivalents of a solute per liter of solution. It is commonly used in acid-base titrations and redox reactions, where the equivalent weight is the mass of a substance that will donate or accept one mole of hydrogen ions or electrons in a reaction.
Additional Knowledge:
·
Molar Mass / Basicity: This refers to a different concept related to molarity and does not define normality.
·
Weight of Compound / Equivalent weight: This expression is closer to the definition of molarity, not normality.
·
Molar mass of the solute / Mass of the solvent: This is used in determining the concentration in terms of molality, not normality.
Top of Form
Bottom of Form





 English
English 150 Questions
150 Questions 150 Mins
150 Mins