Correct option is B
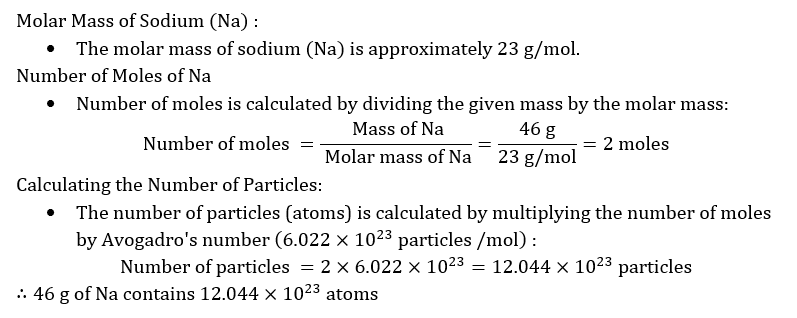
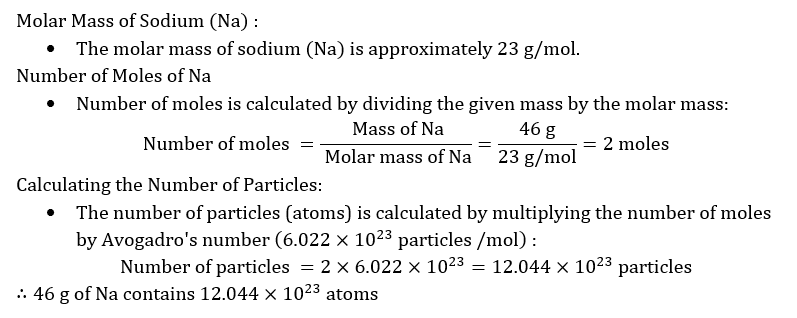
Which law is shown by fixed 1:8 mass ratio in water formation?
Given below are two statements:
Statement I: In preference to a nitrogen-oxygen mixture (present in air), a helium-oxygen mixture is used by divers in deep sea because helium is less soluble in blood than nitrogen at high pressure under the sea.
Statement II: In preference to a nitrogen-oxygen mixture (present in air), a helium-oxygen mixture is used by divers in deep sea because it can readily mix with oxygen than nitrogen.
In the light of the above statements, choose the correct answer from the options given below:
Solubility of CH₃COOH decreases in presence of CH₃COONa due to:
Which of the following foods would you advise to neutralise excess stomach acids?
There are two gas parcels of equal volume, A and B at the same temperature and pressure. Parcel A is one mole of water vapour, while parcel B is one mole of day air. Which of the following is TRUE?
The number of atoms in exactly 12 g of carbon-12 is known as:
Which of the following statements best describes isomerism in chemistry?
Substances like phosphorus burns in air at room temperature. This type of combustion in which a material suddenly bursts into flames, without application of any apparent cause, is called _____________
The present system of atomic masses is based on _________ as the standard.

