Correct option is B
The knocking tendency of hydrocarbons depends on their structure. The higher the knocking tendency, the lower the octane number of the hydrocarbon. Let's analyze the knocking tendencies:
1. n-Alkanes (C):
· Straight-chain alkanes have the highest knocking tendency due to their lower octane number.
2. Branched Alkanes (B):
· Branching in hydrocarbons reduces the knocking tendency because it increases the octane number.
3. Alkenes (D):
· Alkenes exhibit intermediate knocking tendencies due to the presence of double bonds, which increase reactivity.
4. Cycloalkanes (A):
· Cycloalkanes show lower knocking tendencies than alkenes but higher than aromatics due to their stable ring structure.
5. Aromatics (E):
· Aromatic compounds have the lowest knocking tendency among hydrocarbons because of their high octane number and stable delocalized π-electron system.
Order of Decreasing Knocking Tendency:
n-Alkanes > Branched Alkanes > Alkenes > Cycloalkanes > Aromatics
Information Booster: 1. Octane Number:
· A measure of a fuel's resistance to knocking.
· Lower octane numbers correspond to higher knocking tendencies.
2. Factors Affecting Knocking Tendency:
· Chain Structure: Straight chains knock more than branched chains.
· Double Bonds: Increase reactivity and knocking.
· Ring Stability: Cycloalkanes and aromatics are more stable, reducing knocking.
3. Practical Application:
· Fuels with lower knocking tendencies (higher octane numbers) are preferred in high-compression engines.
Additional Knowledge: · n-Alkanes (C):
· Straight-chain hydrocarbons with a high knocking tendency.
· Common examples include propane and hexane.
· Branched Alkanes (B):
· Iso-octane is a benchmark for high-octane fuels due to its branched structure.
· Cycloalkanes (A):
· Examples: Cyclohexane, cyclopentane.
· Moderate knocking tendency due to their stable ring structure.
· Alkenes (D):
· Examples: Ethene, propene.
· Reactive due to double bonds, contributing to intermediate knocking.
· Aromatics (E):
· Examples: Benzene, toluene.
· High stability due to resonance, resulting in low knocking tendency.





 English
English 10 Questions
10 Questions 12 Mins
12 Mins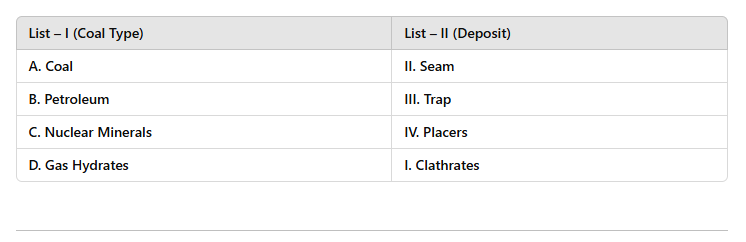 Choose the correct answer:
Choose the correct answer:


