Correct option is C
The given reaction is the combustion of butane (C4H10). Let’s balance it step-by-step:
Step 1: Write the unbalanced reaction:
C4 H10 + Air (O2 + N2) → CO2 + H2O + N2
Step 2: Balance carbon (C):
C4H10 → 4CO2 Thus, 4 moles of CO2 are produced.
Step 3: Balance hydrogen (H):
C4H10 → 5H2O Thus, 5 moles of H2O are produced.
Step 4: Balance oxygen (O):
The total oxygen required = (4 × 2) + (5 × 1) = 13 atoms of oxygen. Since oxygen comes as O2, 13/2 = 6.5 moles of O2 are required.
Step 5: Include nitrogen (N):
Air contains approximately 78% nitrogen and 21% oxygen by volume. For every 1 mole of O2, approximately 3.76 moles of N2 are present. Thus, for 6.5 moles of O2_22: N2 = 6.5 × 3.76 = ~24.5 moles of N2.
Final Balanced Equation:
C4H10 + 6.5O2 + 24.5N2→ 4CO2 + 5H2O + 24.5N2.
Information Booster:
1. Key Stoichiometric Products in Combustion of Hydrocarbons:
· Hydrocarbons combust in the presence of oxygen to produce CO2, H2O, and unused nitrogen from air.
2. Air Composition and its Role in Combustion:
· Air is composed of approximately 21% oxygen and 78% nitrogen. This explains the inclusion of nitrogen in combustion equations.
3. Incomplete Combustion:
· If there isn’t enough oxygen, incomplete combustion occurs, producing CO and soot instead of CO2.
4. Energy Production:
· Combustion of butane is highly exothermic, releasing energy primarily as heat.





 English
English 10 Questions
10 Questions 12 Mins
12 Mins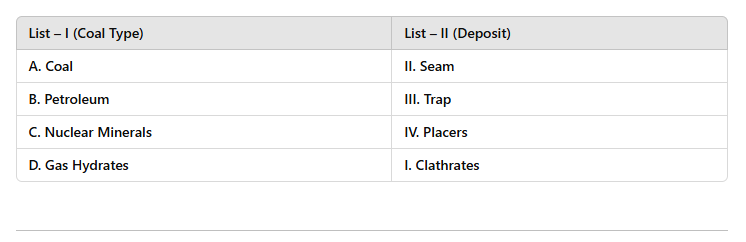 Choose the correct answer:
Choose the correct answer:


