Correct option is D
The correct answer is (d) I < II < III.
Solutions:
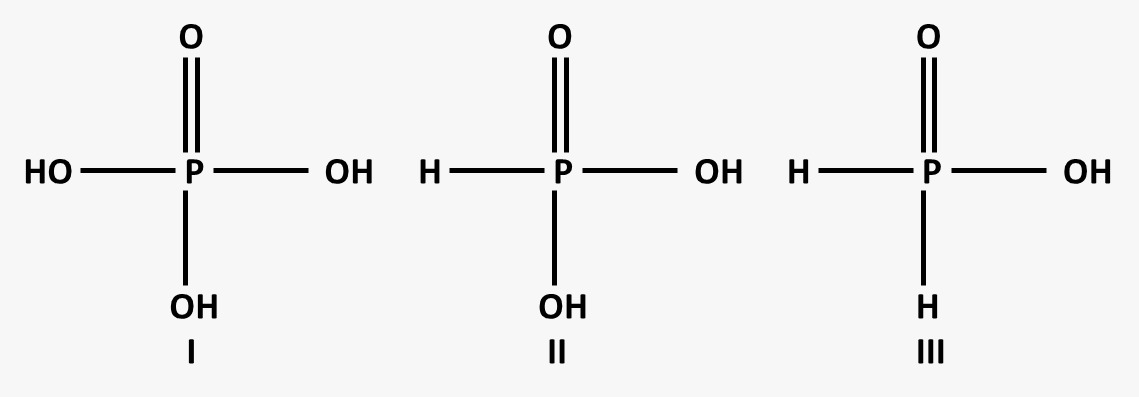
The three acids shown in the image correspond to:
- H₃PO₂ (Hypophosphorous acid)
- H₃PO₃ (Phosphorous acid)
- H₃PO₄ (Phosphoric acid)
- The acidity of oxyacids of phosphorus depends on the number of ionizable -OH groups (hydroxyl groups).
- More electronegative oxygen atoms increase the stability of the conjugate base, making it easier for the acid to lose a proton (H⁺), thereby increasing acidity.
H₃PO₂ (Hypophosphorous acid) → Weakest Acidic
- Contains one -OH group (only one ionizable proton).
- Least tendency to donate H⁺.
H₃PO₃ (Phosphorous acid) → Moderately Acidic
- Contains two -OH groups (two ionizable protons).
- More acidic than H₃PO₂.
H₃PO₄ (Phosphoric acid) → Strongest Acidic
- Contains three -OH groups (three ionizable protons).
- Most acidic among the three.
Since acidic strength increases with the number of ionizable -OH groups, the correct increasing order is:
H3PO2<H3PO3<H3PO4\mathbf{H₃PO₂ < H₃PO₃ < H₃PO₄}H3PO2<H3PO3<H3PO4
Which matches Option (D): I < II < III.





 English
English 100 Questions
100 Questions 90 Mins
90 Mins

