Correct option is B
The correct answer is (b) It shows the properties of its constituents and can have variable composition.
Explanation:
· An alloy is classified as a
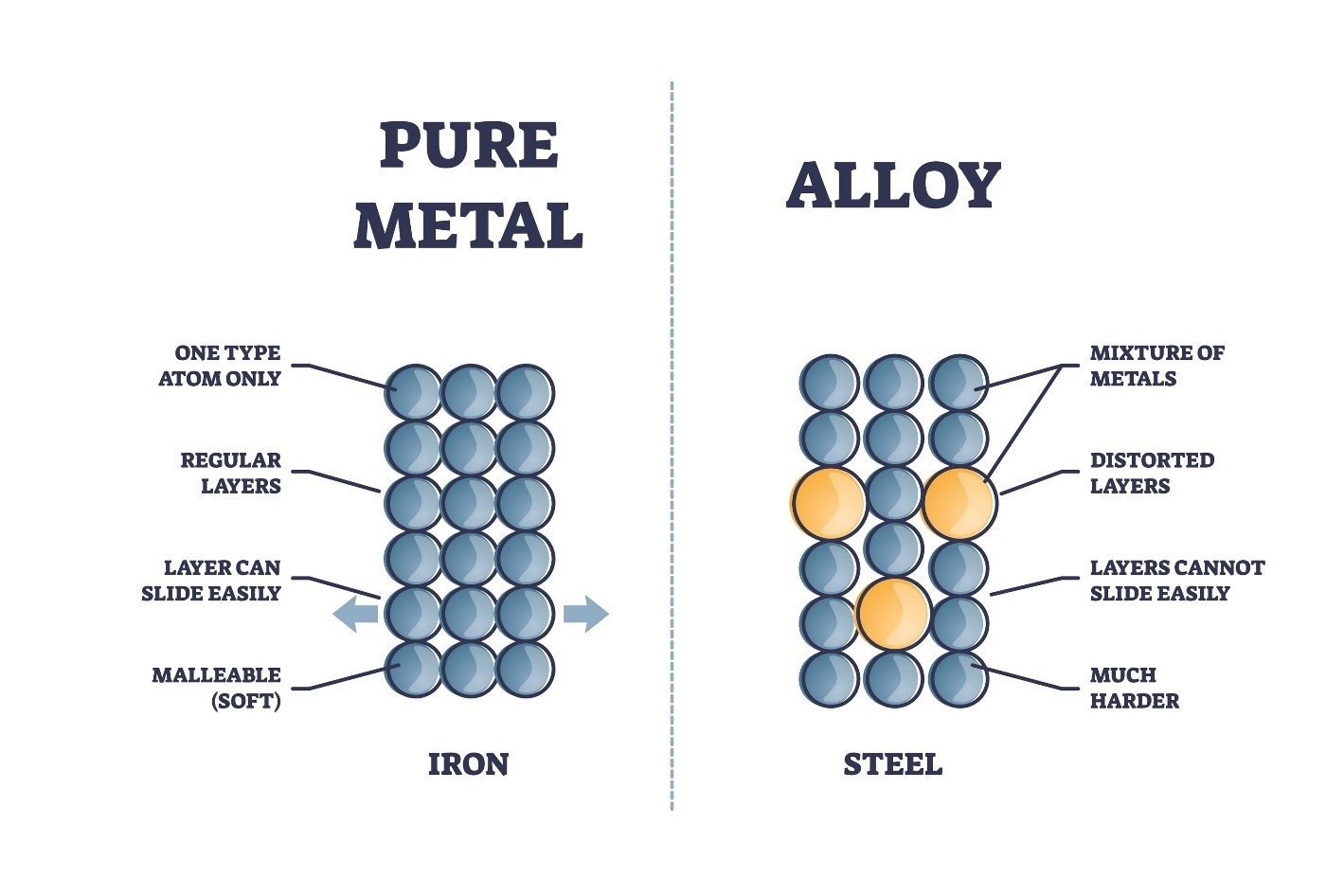
mixture because its constituent substances are not chemically combined, meaning they retain their individual properties.
· Alloys exhibit two key characteristics of mixtures:
·
Variable Composition: The components can be mixed in different ratios (e.g., the proportion of copper to zinc in brass can be adjusted).
·
Retention of Properties: The alloy retains the general properties of the original metallic components (e.g., steel is still metallic and conductive).
 Information Booster:
Information Booster:
· Alloys are generally categorized as
homogeneous mixtures or
solid solutions because the components are uniformly distributed at the atomic level.
· Alloys are created to enhance properties like strength, hardness, or corrosion resistance that pure metals often lack.
Additional Knowledge:
· A chemical
compound is fundamentally different from an alloy because it has a fixed ratio of elements, and the resulting substance has properties distinct from the elements it contains (e.g., water from hydrogen and oxygen).
· The fact that alloys cannot be separated by simple physical methods is due to the atomic-level mixing, but this doesn't change their fundamental classification as mixtures.





 English
English 100 Questions
100 Questions 90 Mins
90 Mins

