Correct option is C
Explanation-
Given:
A protein coding mRNA of 275 amino acids (~30 kDa) with 3 methionines at - Position 1, Position 135 and Position 230
This protein is alanine-rich (~35%) and tagged with a 6xHis tag at the C-terminal for purification. Translation was initiated in vitro and supplemented with radioactive ¹⁴C-Ala at 5 minutes.
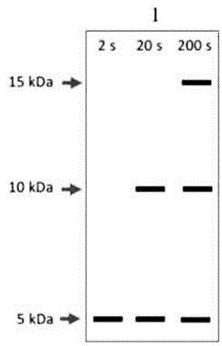
Aliquots were taken at 2s, 20s, 200s, translation stopped, protein purified, and digested with CNBr (cleaves at methionine). Fragments generated by CNBr are ~15 kDa, ~10 kDa, and ~5 kDa.
CNBr (Cyanogen Bromide) cleaves C-terminal to methionine residues.
So a protein with Met at 1, 135, 230 will be cut into:
Residues 1–134 (~15 kDa)
135–229 (~10 kDa)
230–275 (~5 kDa)
Pulse-labeling with ¹⁴C-Ala after 5 min:
Labels only newly synthesized proteins.
The earlier the timepoint, the less complete translation.
SDS-PAGE shows only ¹⁴C-labeled fragments.
By Analyze the Timepoints:
At 2 seconds: Only short peptides are made → the ribosome likely hasn’t gone far beyond the first ~40–50 amino acids.
After CNBr cleavage, only the first fragment (15 kDa) may show partial labeling, or no fragment fully labeled.
At 20 seconds: Enough time for elongation up to second or third methionine.
Expect partial translation of longer fragments.
Possibly labeling of 10 kDa and 5 kDa regions.
At 200 seconds: Full-length protein synthesized.
All three CNBr fragments can appear strongly labeled.
Expected Autoradiogram Pattern:
2 sec → Weak labeling, possibly 5 kDa only or none strongly visible.
20 sec → Moderate labeling of 5 kDa, and maybe 10 kDa bands.
200 sec → Strong labeling of all 3 bands (15, 10, 5 kDa).
Option C is the correct answer because it accurately reflects the time-dependent synthesis and labeling of the CNBr-cleaved protein fragments, as visualized via autoradiography.
2 sec: Faint or single lower band (incomplete protein)
20 sec: Multiple bands (partial labeling of intermediate-length proteins)
200 sec: All bands clearly visible (fully translated and labeled proteins)