Correct option is D
Given:
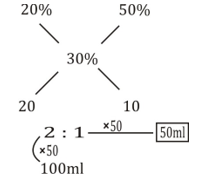
A 100 ml solution of H2SO4 having concentration of 20% is mixed with a 50% concentrated x ml mixture
Such that the net mixture is 30% concentrated
Concept used:
C1V1 + C2V2 = C3V3
Solution:
According to the question,
(100 × 20) + (50 × x) = 30 × (100 + x)
=> 2000 + 50x = 3000 + 30x
=> 50x - 30x = 3000 - 2000
=> 20x = 1000
=> x = 1000/20 = 50 ml
∴ The value of x is 50 ml.
Alternative Method:





 English
English 100 Questions
100 Questions 60 Mins
60 Mins

