Correct option is B
Sol.
The gel filtration shows a 150 kDa species, indicating a complex or multimer.
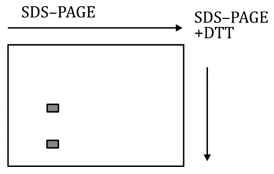
In SDS-PAGE without DTT, the complex remains intact, showing one or few bands corresponding to subunits linked by covalent bonds (disulfide bridges).
Upon SDS-PAGE with DTT, disulfide bonds are reduced, and the protein complex dissociates into smaller subunits, visible as separate bands.
This implies covalent linkage (disulfide bonds) between subunits, confirming multiple proteins linked covalently.
Information Booster:
DTT (dithiothreitol) reduces disulfide bonds, disrupting covalent linkages.
Non-covalent interactions would be disrupted by SDS alone, so complexes linked non-covalently wouldn't maintain structure in SDS-PAGE without DTT.
2D SDS-PAGE with and without reducing agents helps identify protein complexes and subunit structure.