Correct option is A
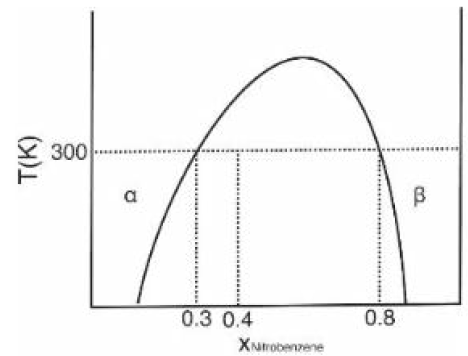
The phase diagram shows the equilibrium composition of the two phases, α (hexane-rich) and β (nitrobenzene-rich), at 300 K.
The lever rule is a formula used to determine the mole fraction (xi) of each phase of a binary equilibrium phase diagram. It can be used to determine the fraction of liquid and solid phases for a given binary composition and temperature that is between the liquidus and solidus line.
The total moles of mixture = 0.6 mol hexane + 0.4 mol nitrobenzene = 1.0 mol.
Let nα and nβ be the number of moles in the α and β phases, respectively.
Using the lever rule: