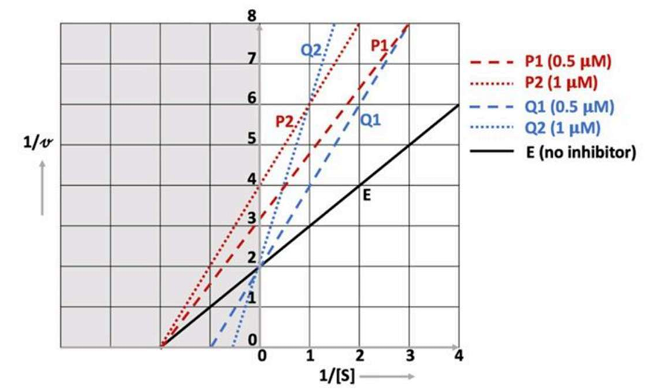
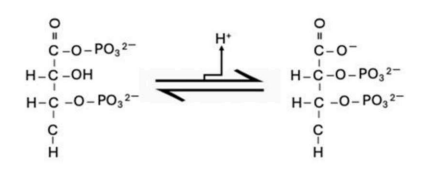
Correct option is A
Step 1: Convert distance to nm
r = 2.9 Å = 0.29 nm
Step 2: Calculate charge product
q₁ = +0.25e
q₂ = –0.35e
Therefore,
q₁ × q₂ = –0.0875 e²
Step 3: Electrostatic energy for one bond
Formula:
E = (k × q₁ × q₂) / (D × r)
Difference in dielectric
ΔE = (k × q₁ × q₂ / r) × (1/D_core – 1/D_water)
Substitute values:
ΔE = (1389 × (–0.0875) / 0.29) × (1/4 – 1/78.5)
Step-by-step check:
1389 × 0.0875 = 121.5375
Divide by 0.29 → 419.1
(1/4 – 1/78.5) = (0.25 – 0.01274) = 0.23726
Multiply 419.1 × 0.23726 ≈ 99.5
Apply negative sign (since q₁ × q₂ is negative) → ΔE (for one bond) ≈ –99.5 kJ/mol
For 3 bonds:
ΔE_total = 3 × (–99.5) = –298.5 kJ/mol
Final Answer:
The total electrostatic energy difference for 3 bonds ≈ –298.5 kJ/mol