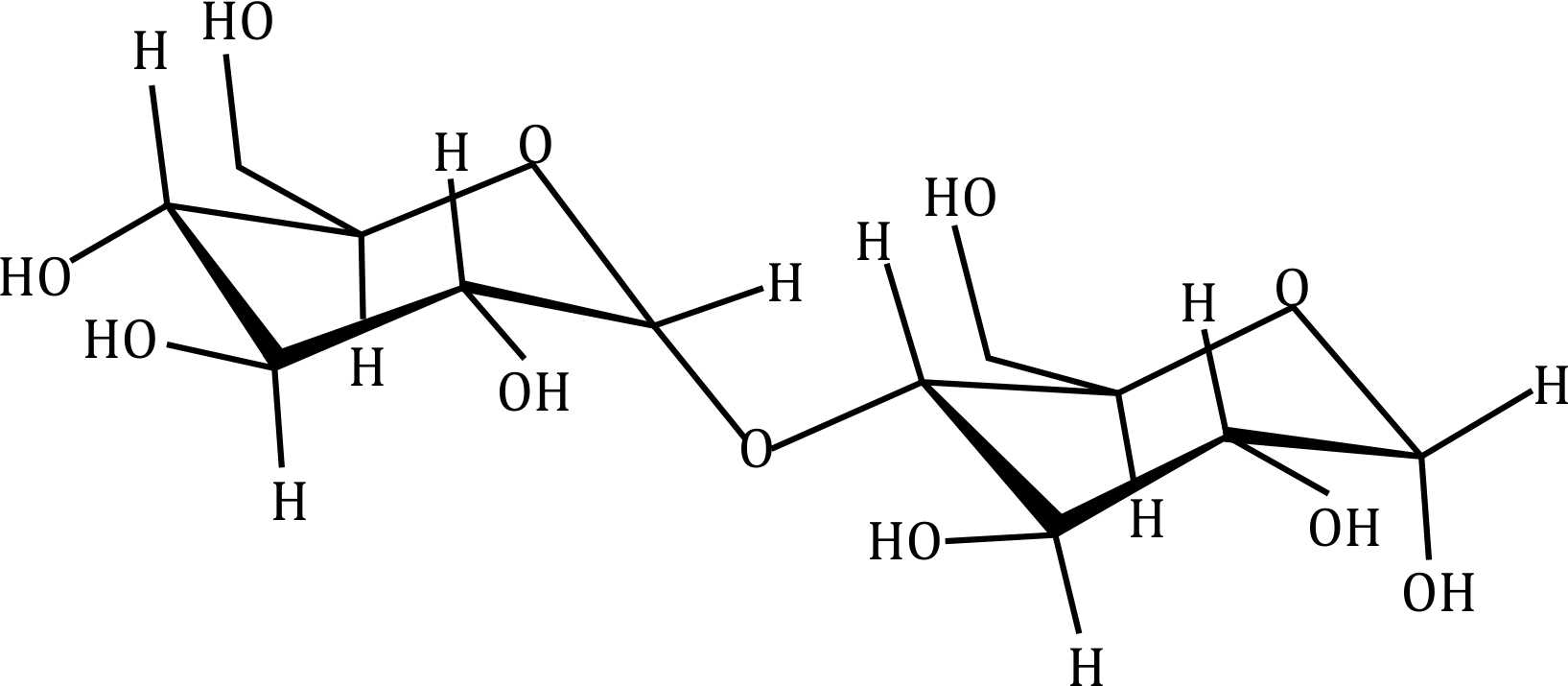
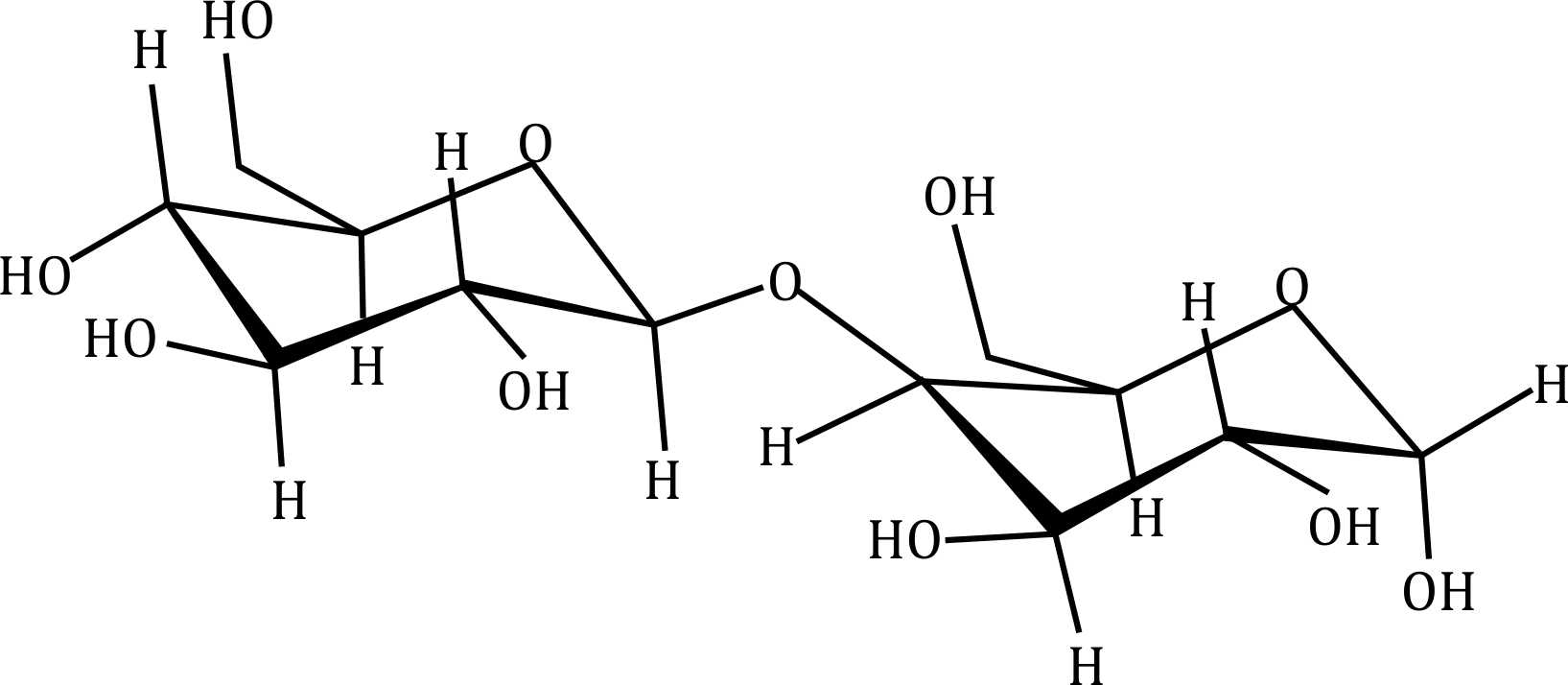
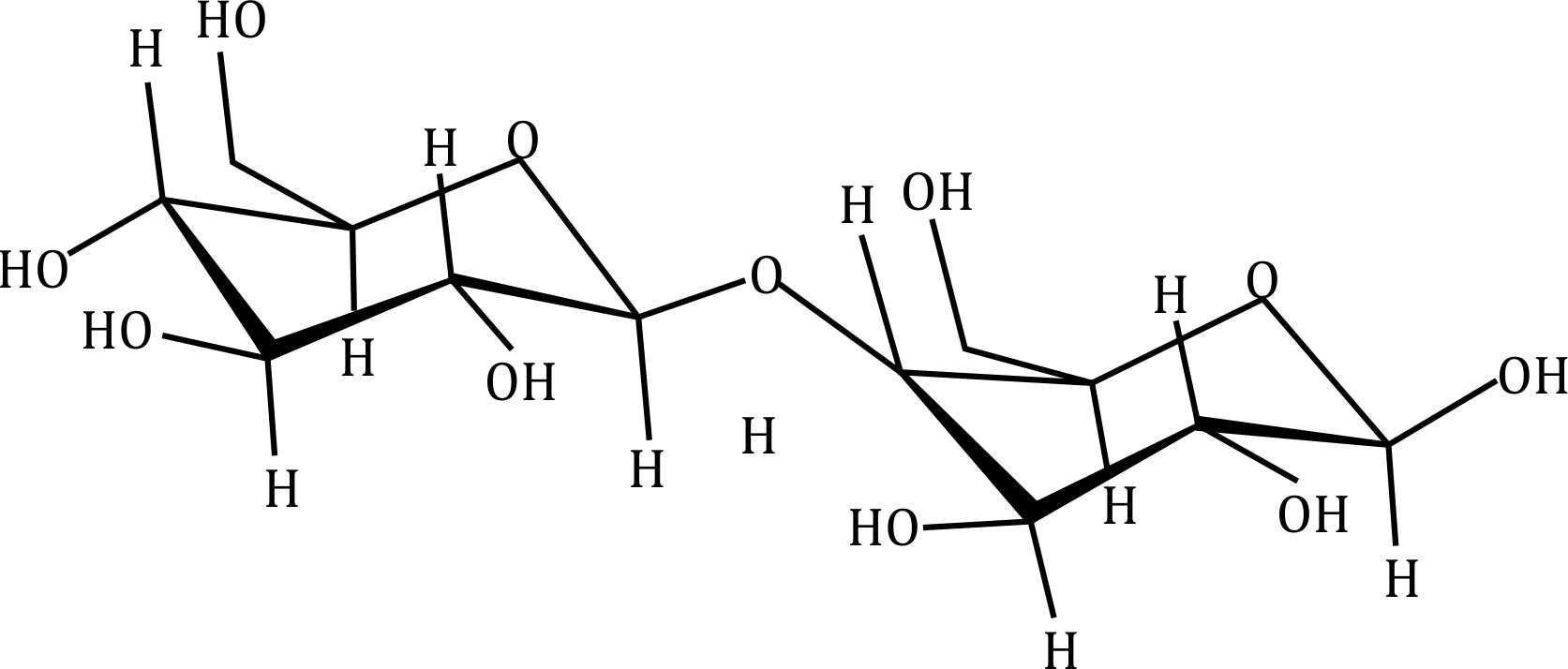
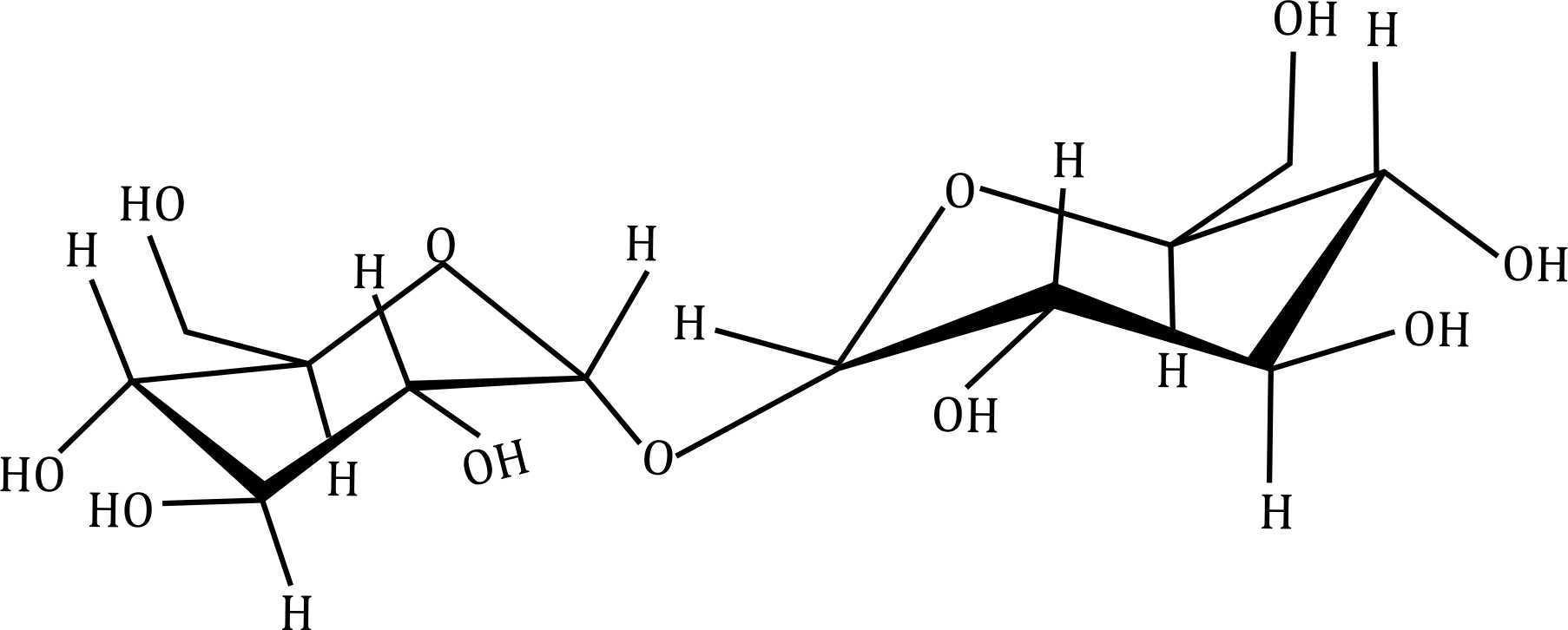
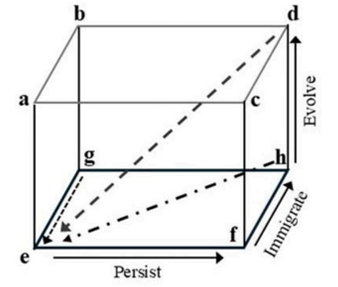
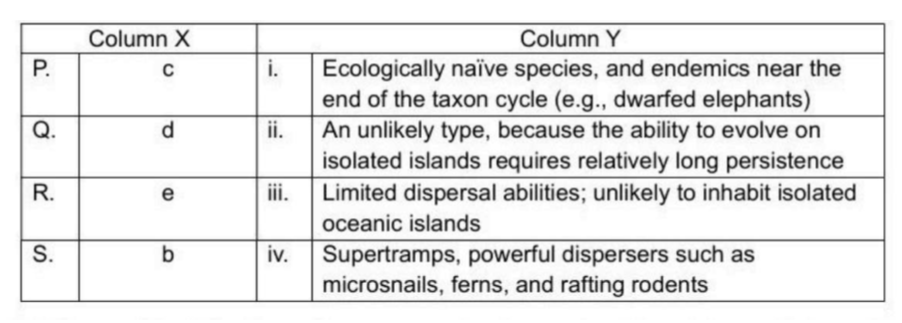
Correct option is D
To determine which sugar will not reduce Tollen's reagent, we need to analyze whether the sugar is a reducing or non-reducing sugar.
- Reducing sugars: These sugars have a free anomeric carbon (with a free aldehyde or ketone group) that can reduce Tollen’s reagent (Ammoniacal Silver Nitrate, AgNO₃/NH₃), leading to the formation of a silver mirror.
- Non-reducing sugars: These sugars do not have a free anomeric carbon and cannot reduce Tollen's reagent. Typically, disaccharides like sucrose fall into this category because their glycosidic bond involves the anomeric carbons of both monosaccharide units.
Step 1: Identifying the Structures
Looking at the structures:
- Option (a): Appears to be a disaccharide with a free anomeric carbon.
- Option (b): Another disaccharide, possibly lactose or maltose, which are reducing sugars.
- Option (c): Seems to be a trisaccharide, which could be maltotriose.
- Option (d): Appears to be sucrose, which is a non-reducing sugar.
Step 2: Understanding Sucrose
Sucrose is a non-reducing sugar because:
- It is composed of glucose and fructose linked by a glycosidic bond between their anomeric carbons (α-1,2 linkage).
- This bond locks both anomeric carbons, preventing them from opening into their aldehyde or ketone forms.
- Since there is no free anomeric carbon, it cannot reduce Tollen’s reagent.
Final Answer:
The sugar that will not reduce Tollen's reagent is sucrose in option (d).
Information Booster
- Reducing sugars contain a free aldehyde (-CHO) or ketone (-C=O) functional group that can be oxidized.
- Tollen’s test uses ammoniacal silver nitrate (AgNO₃ + NH₃) to detect reducing sugars. A silver mirror forms if the sugar is reducing.
- Common reducing sugars: Glucose, fructose, galactose, maltose, lactose.
- Sucrose is a non-reducing sugar because it has an α-1,2 glycosidic bond, locking both anomeric carbons.
- Disaccharides like maltose and lactose are reducing sugars because one anomeric carbon remains free.
- Polysaccharides (starch, cellulose) do not react with Tollen’s reagent because their glycosidic bonds prevent oxidation.
- Barfoed’s test is another test for reducing sugars, distinguishing monosaccharides from disaccharides.