Correct option is B
Explanation-
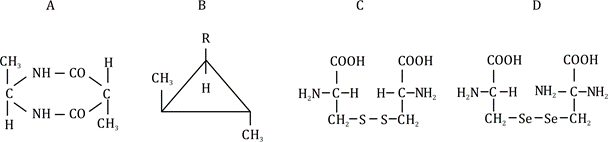
Molecule A: The structure is a five-membered ring with two -CO groups and three -CH₃ groups. The carbon atoms in the ring are each bonded to two other carbons in the ring, a -CO or -CH₃ group, and a hydrogen (implied). However, the molecule has a plane of symmetry through the ring (due to the symmetrical arrangement of the -CH₃ and -CO groups). Thus, it is not optically active.
Molecule B: This is a triangular three-membered ring with a -CH₃ group on one carbon and an -R group on another. The carbon with the -CH₃ group is bonded to two other carbons in the ring, a -CH₃, and a hydrogen, making it a chiral center (four different groups). The carbon with the -R group also has four different groups (two ring carbons, -R, and H). The molecule lacks a plane of symmetry, so it is optically active.
Molecule C: This is a straight chain with a -COOH group at one end, a central carbon bonded to -NH₂ and -H, and a -CH₂-CH₂-CH₃ group. The central carbon is bonded to -H, -NH₂, -COOH (via the chain), and -CH₂-CH₂-CH₃. These are four different groups, making it a chiral center. The molecule lacks a plane of symmetry, so it is optically active.
Molecule D: This is similar to C, but the central carbon is bonded to -Se- instead of -S-. The central carbon is still bonded to four different groups: -H, -NH₂, -COOH (via the chain), and -CH₂-Se-CH₃. This makes it a chiral center, and the molecule lacks a plane of symmetry, so it is optically active.
Conclusion:
Molecule A is not optically active.
Molecules B, C, and D are optically active
Final Answer: Based on the analysis, the molecules B, C, and D are optically active, but this does not match any option. If choose, option 2 (B and D only) is the closest so that makes it a correct answer.