Correct option is D
The correct answer is (d) Only I.
Explanation:
1. Reaction I: 2Ag⁺ (aq) + 2e⁻ → 2Ag (s) This reaction is a reduction reaction because the silver ions (Ag⁺) gain electrons (2e⁻) to form solid silver (Ag). In a reduction reaction, there is a gain of electrons, which decreases the oxidation state of the species involved. Here, Ag⁺ is reduced to Ag.
2. Reaction II: Cu (s) → Cu²⁺ (aq) + 2e⁻ This reaction is an oxidation reaction, not a reduction reaction. In this process, solid copper (Cu) loses electrons (2e⁻) to form copper ions (Cu²⁺), increasing the oxidation state of copper from 0 to +2. Therefore, this is an oxidation reaction rather than a reduction reaction.
Conclusion:
· Only Reaction I is a reduction reaction as it involves the gain of electrons by Ag⁺.
· Reaction II is an oxidation reaction as it involves the loss of electrons by Cu.





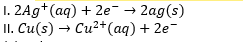
 English
English 10 Questions
10 Questions 10 Mins
10 Mins

