Correct option is A
Ans.(a)
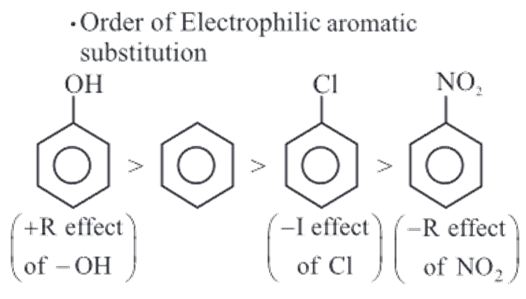
Greater is the electron density on benzene ring, greater is the reactivity towards electrophilic aromatic substitution.
Electron withdrawing group like NO2,Cl decreases the electron density on benzene group.
Phenol is more reactive than benzene towards electrophilic substitution reaction. The donation of the oxygen’s lone pair into the ring system increases the electron density around the ring. That makes the ring much more reactive than it is in benzene. The intermediate carbocation is more resonance stabilized.









 English
English 200 Questions
200 Questions 200 Mins
200 Mins




