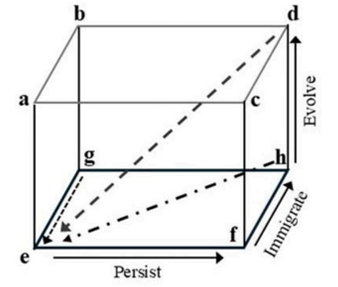
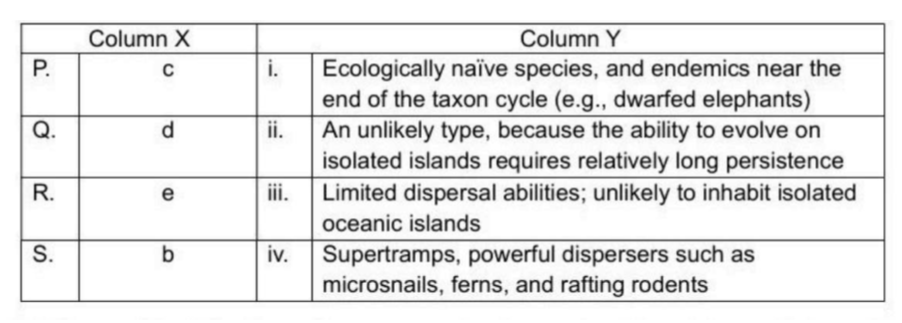
Correct option is A
Explanation-
Given pKa values:
α-amino group (N-terminus): pKa = 9.6
α-carboxyl group (C-terminus): pKa = 2.3
Tyrosine (Tyr) side chain –OH: pKa = 10.46
Arginine (Arg) side chain –NH₃⁺: pKa = 12.48
Step-by-step Analysis at pH = 5.0:
1. N-terminal amino group (pKa = 9.6):
pH 5.0 < pKa → Protonated → +1 charge
2. C-terminal carboxyl group (pKa = 2.3):
pH 5.0 > pKa → Deprotonated → –1 charge
3. Tyrosine side chain (pKa = 10.46):
pH 5.0 < pKa → Protonated → neutral (–OH group not ionized)
4. Arginine side chain (pKa = 12.48):
pH 5.0 < pKa → Protonated → +1 charge
Total net charge:
N-terminal (α-NH₃⁺) : +1
C-terminal (–COO⁻) : –1
Tyrosine side chain (–OH) : 0
Arginine side chain (–NH₃⁺) : +1
Total +1
Final Answer:
Option a : 1.0