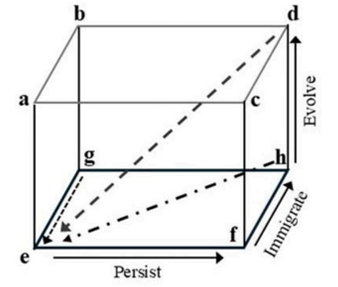
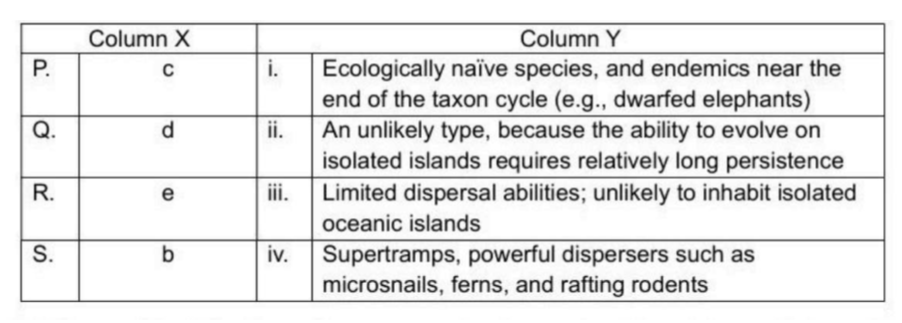
Correct option is C
The change in the S value from 1.83 (monomeric state) to 2.57 (after 2 days) indicates an increase in the size and shape of the enzyme, which suggests that it has dimerized (formed a homodimer). The S value is indicative of the sedimentation rate, and as molecules form larger complexes (like dimers, trimers, etc.), their sedimentation rate increases. This change in the S value from the monomeric to the dimeric state suggests a monomer to homodimer transition.
Information Booster:
The S value is directly related to the size and shape of a molecule, with larger molecules sedimenting faster.
A monomeric enzyme has a specific S value, and when it transitions to a dimer, the increased size leads to a higher S value.
Homodimers are composed of two identical subunits, and the transition from monomer to dimer is common in enzymes.
Ultracentrifugation is a technique used to measure the sedimentation velocity of proteins and can provide insight into their size, shape, and conformation.
The transition from monomer to dimer can also be influenced by buffer conditions and time, which may explain the change observed after 2 days.
This phenomenon is important in enzyme activity, as dimerization can sometimes activate the enzyme or influence its function.
Additional Knowledge:
Option 1: "The enzyme is aggregated and forms a homo-octamer" is incorrect because the observed change in the S value suggests a dimerization, not an aggregation into an octamer.
Option 2: "The enzyme is monomeric but is now associated with the buffer components" would not explain the increase in the S value; the association with buffer components would typically result in little or no change in the enzyme's size.
Option 4: "The enzyme has degraded due to autocatalytic activity" is incorrect because degradation would typically lead to a decrease in the S value, not an increase.