Correct option is A
A thermometric titration is one of a number of instrumental titration techniques where endpoints can be located accurately and precisely without a subjective interpretation on the part of the analyst as to their location. Enthalpy change is arguably the most fundamental and universal property of chemical reactions, so the observation of temperature change is a natural choice in monitoring their progress.
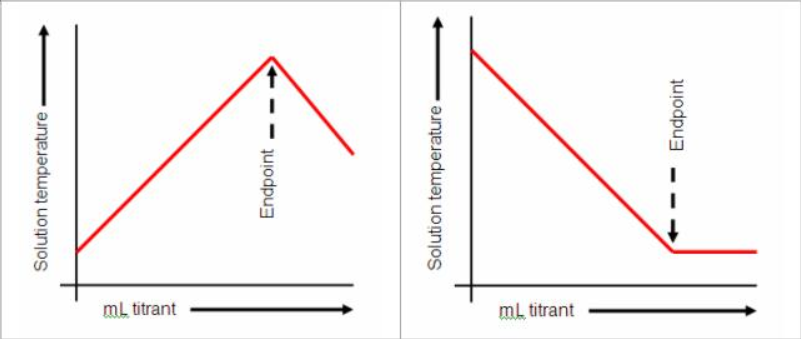
In the thermometric titration, titrant is added at a known constant rate to a titrand until the completion of the reaction is indicated by a change in temperature. The endpoint is determined by an inflection in the curve generated by the output of a temperature measuring device.
Consider the titration reaction:
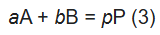
Where:
A = the titrant, and a = the corresponding number of moles reacting
B = the analyte, and b = the corresponding number of moles reacting
P = the product, and p = the corresponding number of moles produced
At completion, the reaction produces a molar heat of reaction ΔHr which is shown as a measurable temperature change ΔT. In an ideal system, where no losses or gains of heat due to environmental influences are involved, the progress of the reaction is observed as a constant increase or decrease of temperature depending respectively on whether ΔHr is negative (indicating an exothermic reaction) or positive (indicating an endothermic reaction). In this context, environmental influences may include (in order of importance):
Heat losses or gains from outside the system via the vessel walls and cover;
Differences in the temperature between the titrant and the titrand;
Evaporative losses from the surface of the rapidly mixed fluid;
Heats of solution when the titrant solvent is mixed with the analyte solvent;
Heat introduced by the mechanical action of stirring (minor influence); and
Heat produced by the thermistor itself (very minor influence).
The Gibbs free energy change (ΔGo) is related to the change in enthalpy (ΔHo), change in entropy (ΔSo) and temperature (T) according to the equation:
ΔG°= ΔHo-TΔSo.
For a spontaneous process: ΔG < 0.For a thermometric titration more high value of IΔHI indicates more high value of IΔGI and ΔG < 0.







