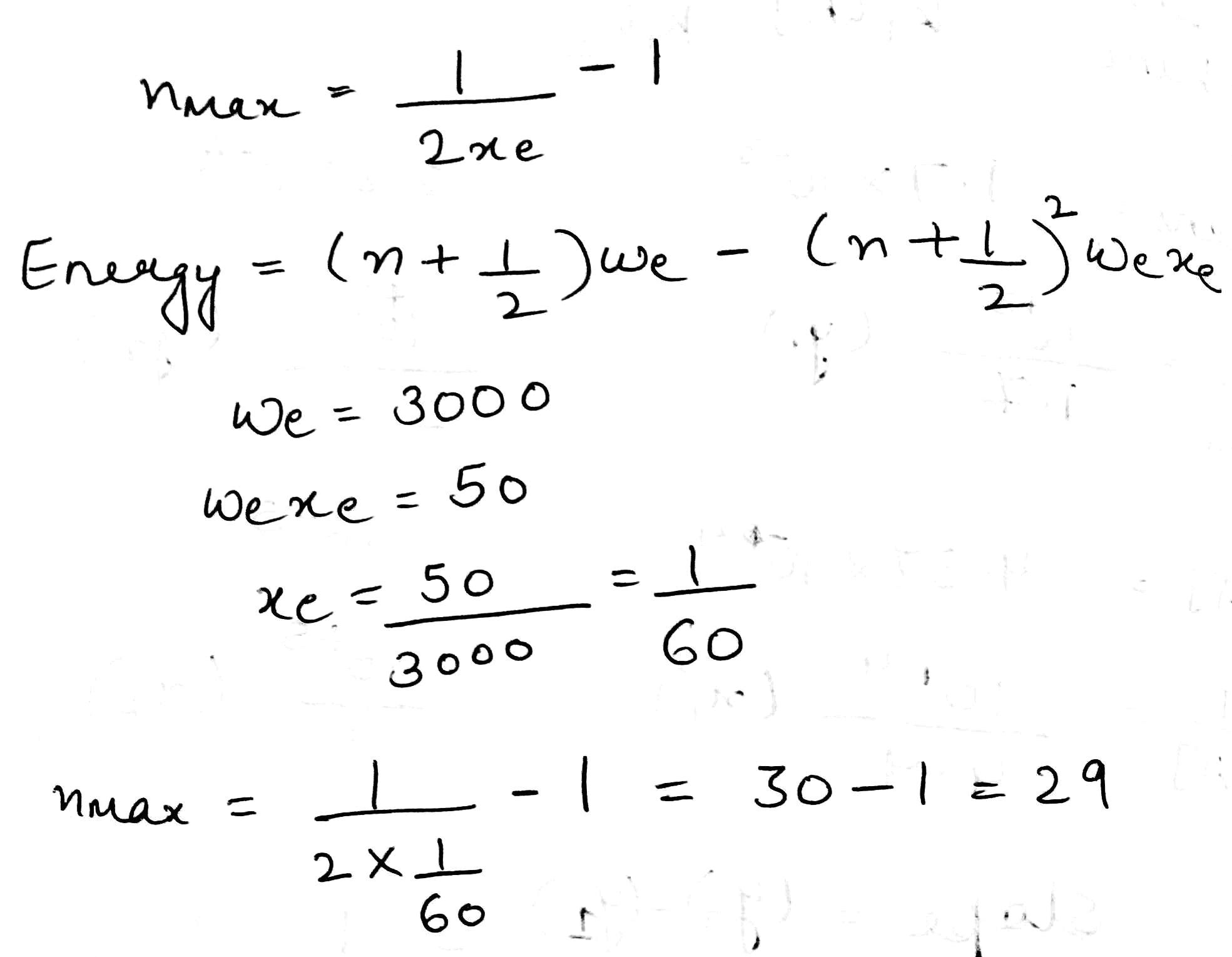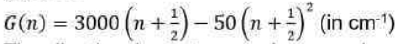Correct option is A
Vibrational spectra of diatomic molecules
A diatomic molecule with atomic masses m1 and m2 joined by a chemical bond vibrates as a one-dimensional simple harmonic oscillator (SHO). Classically, the vibrational frequency of a mass point m connected by a spring of force constant k is given by
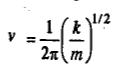
In the case of a diatomic molecule, the masses m1 and m2 vibrate back and forth relative to their centre of mass in opposite directions
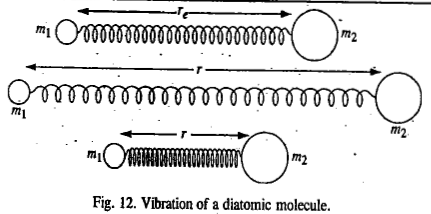
The two masses reach the extreme of their respective motions at the same time. The vibrational frequency of the molecule is given by:
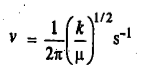
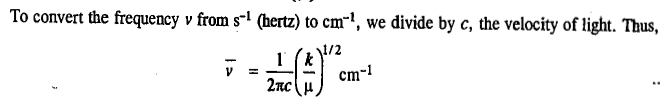
The units of the force constant are Nm-1. The potential energy of the simple harmonic oscillator (SHO) as a function of displacement from the equilibrium configuration is given by the parabolic Hooke's law equation
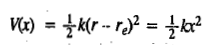
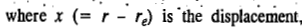
re being the equilibrium bond length
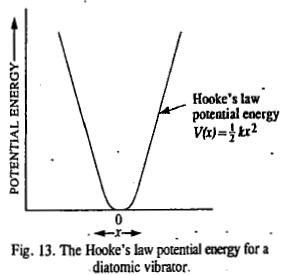


At room temperature, most of the molecules are in the ground vibrational state (v=0) so that the only transition of interest is that which takes place from v=0 to v=1. The vibrational frequency corresponding to this transition is called fundamental vibrational frequency. Diatomic molecules have only one vibrational frequency. This is a stretching vibrational frequency. Diatomic molecules do not have a bending vibrational frequency.
Vibrational spectra are observed in the infrared (IR) region. There is a very important requirement for a molecule to show an infrared spectrum. It states that the dipole moment of the molecule must change during the vibration. Homonuclear diatomic molecules such as H2, O2, N2, etc., do not have a permanent dipole moment nor does the stretching of the bond between the two atoms change the dipole moment from zero. Hence, homonuclear diatomics do not show IR spectra. On the other hand, heteronuclear diatomic molecules such as CO, NO, CN, HCl, do possess a dipole moment which changes when the bond length changes. Hence they show IR spectra. Thus, homonuclear diatomics are IR-inactive while heteronuclear diatomics are IR-active.
In practice, the molecule does not always vibrate as a simple harmonic oscillator; there is present what is called anharmonicity. In 1929, P.M. Morse suggested an empirical expression for the potential energy of an anharmonic diatomic oscillator, given by
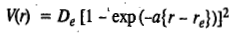
Here a is a constant and De is the dissociation energy of the molecule. The Morse potential energy is sketched as
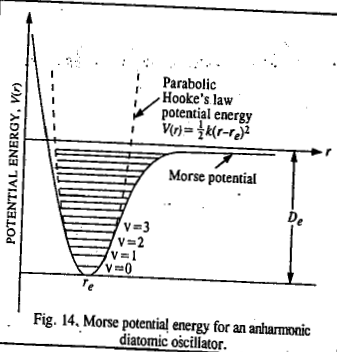
It may be pointed out that the dissociation energy De in the Morse potential energy curve is measured from the bottom of the potential well. The experimentally measured dissociation energy D0 is the one that is measured from the ground level (v=0) to the top.
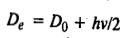
When the Schrödinger equation for an anharmonic oscillator is solved using the Morse potential energy, the energy levels are given by
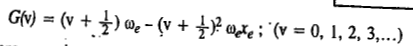
where
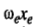
is called the anharmonicity constant. As expected;
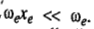
The consequence of anharmonicity is that the vibrational energy levels of the S.H.O. are all slightly lowered and the spacing between them is no longer constant but goes on steadily decreasing with increase in vibrational quantum number. Also, because of anharmonicity, the rule for vibrational transitions is no longer
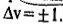
Instead, transitions corresponding to
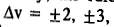
, etc., are also observed in the IR spectra. These are called the first overtone, the second overtone, etc., respectively. The intensity of an overtone is dependent on the anharmonicity of the vibration. Compared with the highly intense fundamental vibrational frequency, the overtones are very weak, i.e., of considerably low intensity.