Correct option is C
The correct answer is (c) s⁻¹.
For a first-order reaction, the rate law is expressed as:
Rate=k[A]
where:
· Rate has units of concentration per time, typically mol L⁻¹ s⁻¹ (or equivalent).
· [A] is the concentration of the reactant, typically in mol L⁻¹.
Rearranging for kkk, the rate constant:
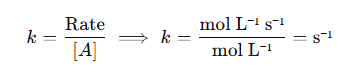
Thus, the unit of kkk for a first-order reaction is s⁻¹.
Information Booster: 1. Reaction Order: The sum of the exponents of the concentration terms in the rate law.
2. First-Order Reaction: The rate depends linearly on the concentration of one reactant.
3. Rate Law: Expresses the relationship between the rate of a reaction and the concentrations of reactants.
4. Unit of kkk:
· Zero-order: mol L⁻¹ s⁻¹
· First-order: s⁻¹
· Second-order: L mol⁻¹ s⁻¹ (or mol⁻¹ cm³ s⁻¹)
5. Half-Life: For a first-order reaction, the half-life is independent of the initial concentration:
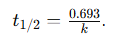
Additional Knowledge: · (a) mol cm⁻³ s⁻¹: Incorrect; this is the unit for the rate of a zero-order reaction.
· (b) mol⁻¹ cm³ s⁻¹: Incorrect; this is the unit for a second-order reaction rate constant.
· (c) s⁻¹: Correct for a first-order reaction.
· (d) mol⁻¹ cm³ s: Incorrect; this does not correspond to any reaction rate constant.





 English
English 10 Questions
10 Questions 12 Mins
12 Mins


