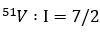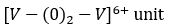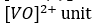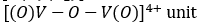Correct option is B
Electron paramagnetic resonance (EPR) spectroscopy (also called electron spin resonance (ESR) spectroscopy), is used to study paramagnetic species with one or more unpaired electrons, e.g. free radicals, diradicals, metal complexes containing paramagnetic metal centres, defects in semiconductors and irradiation effects in solids. While diamagnetic materials are EPR silent, paramagnetic species always exhibit an EPR spectrum. This consists of one or more lines, depending on the interactions between the unpaired electron (which acts as a ‘probe’) and the molecular framework in which it is located. Analysis of the shape of the EPR spectrum (the number and positions of EPR lines, their intensities and line widths) provides information about the paramagnetic species, e.g. the structure of a free radical, characterization of the coordination sphere around the metal centre in a coordination complex, or the presence of multiple paramagnetic species.
We can obtain information about nuclei with nuclear spin quantum number I not equal to 0 which are close to the paramagnetic centre. The spins of such nuclei interact magnetically with the unpaired electron and give rise to a hyperfine interaction.
EPR spectrum of one line is split and the number of lines in the hyperfine pattern is given by 2nI+1, where n is the number of equivalent nuclei with spin quantum number I.
VO2+ (3d1 ) that is oxidovanadium (IV) complexes: The oxidovanadium (IV) complexes i.e.
oxidovanadium (IV) complexes experience a strong Jahn Teller distortion (The Jahn–Teller theorem essentially states that any non-linear molecule with a spatially degenerate electronic ground state will undergo a geometrical distortion that removes that degeneracy, because the distortion lowers the overall energy of the species) and the orbitally singlet ground state is widely separated from the immediate excited electronic state (i.e. energy separation, ΔE >> kBT) and consequently, the spin-lattice relaxation process is disfavoured (i.e. spin- lattice relaxation time T1 is relatively longer).It makes their ESR studies easier and it can be studied even at room temperature. Because of the same ground, the EPR studies of the Cu (II) complexes can be made even at room temperature.
In the monomeric oxidovanadium (IV) complexes, the unpaired electron interacts with the nuclear
spin I= 7/2 of vanadium. It splits the ESR signal into equally spaced 8 hyperfine peaks (2 x 7/2 + 1 = 8)