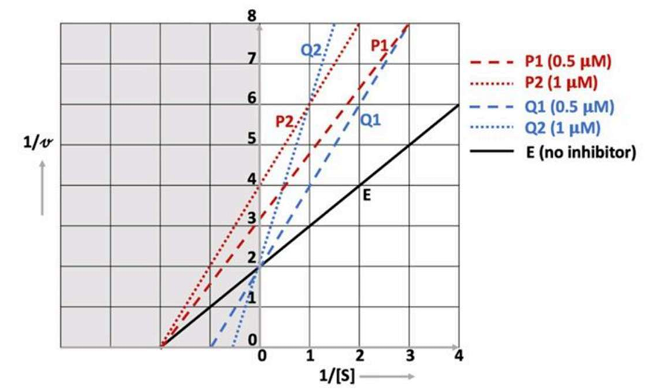
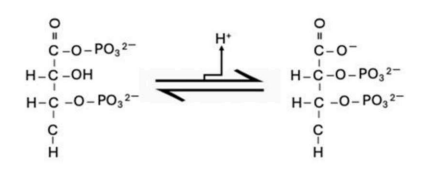
Correct option is D
pH of endocytic vesicle = 5.2
[H⁺] concentration of endocytic vesicle => pH = -log [H⁺]
5.2 = -log [H⁺]
∴ [H⁺] = 6.31 × 10⁻⁶ mol/L
pH of gastric juice = 2
[H⁺] concentration of gastric juice =>
pH = -log [H⁺]
2 = -log [H⁺]
∴ [H⁺] = 1 × 10⁻² mol/L
∴[H⁺] of gastric juice
------------------------------
[H⁺] of endocytic vesicle
= (1 × 10⁻²) / (6.31 × 10⁻⁶)
= (1 / 6.31) × 10⁴
= 0.1585 × 10⁴
= 1585
Information Booster:
- The pH scale is logarithmic; a one-unit change represents a tenfold difference in [H+].
- Gastric juice has a highly acidic environment, aiding in digestion.
- Endocytic vesicles typically maintain a near-neutral to slightly acidic pH.
- A pH difference significantly impacts enzymatic activity and cellular transport mechanisms.
- The ratio between [H+] can be calculated using pH = pH2-pH1