Correct option is C
The Maxwell distribution concerns the distribution of an amount of energy between identical but distinguishable particles. It represents the probability of the distribution of the states in a system having different energies.

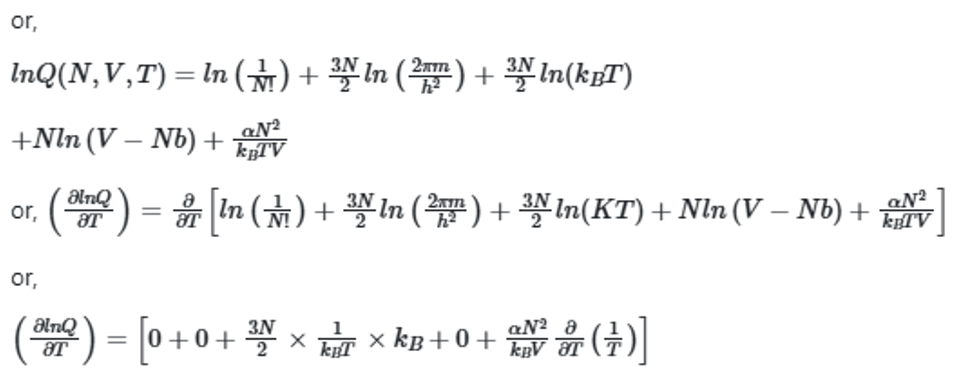
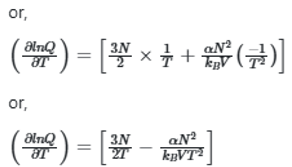
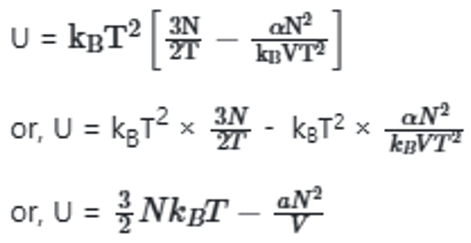
The partition function for a gas is given by
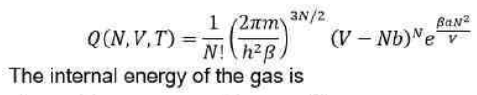
The Maxwell distribution concerns the distribution of an amount of energy between identical but distinguishable particles. It represents the probability of the distribution of the states in a system having different energies.

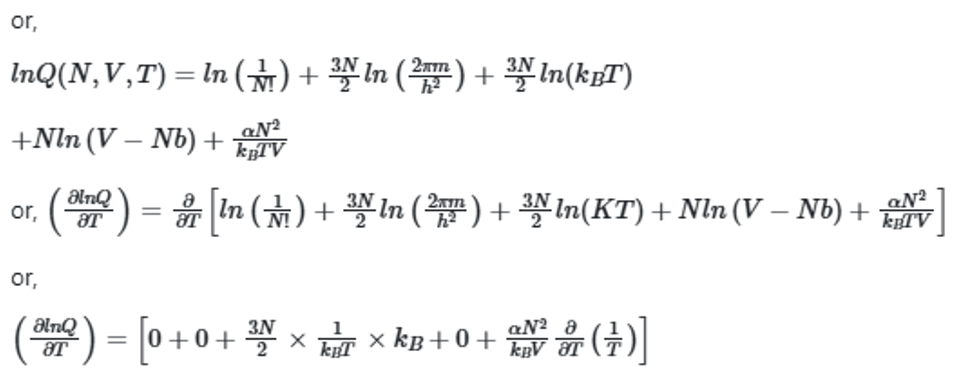
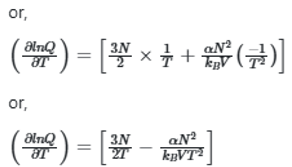
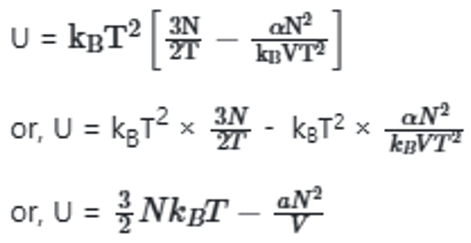
The system with the largest internal pressure is
A compound consists of atoms A and B. Atoms of B form cubic close packing lattice and atoms of A occupy half of octahedral voids and half of tetrahedral voids. What is the molecular formula of the compound?
Entropy associated with a die with all its six faces having equal probability of being on top is (kB : Boltzmann constant):

The equivalent symmetry operations for and are, respectively,
The system with the largest internal pressure is
A compound consists of atoms A and B. Atoms of B form cubic close packing lattice and atoms of A occupy half of octahedral voids and half of tetrahedral voids. What is the molecular formula of the compound?
Entropy associated with a die with all its six faces having equal probability of being on top is (kB : Boltzmann constant):

The equivalent symmetry operations for and are, respectively,

