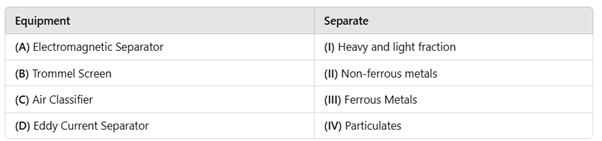
Correct option is B
Polychlorinated Biphenyls (PCBs) consist of
two benzene rings connected by a single carbon-carbon bond. These rings can have up to 10 chlorine atoms substituted at various positions. The general structure of PCBs is C₁₂H₁₀₋ₓClₓ, where x represents the number of chlorine atoms.
The presence of these two benzene rings contributes to the chemical stability and persistence of PCBs, making them resistant to degradation.
Information Booster: 1. PCBs are made of biphenyl molecules, which means two benzene rings joined together.
2. They can have 1 to 10 chlorine atoms substituted on these rings.
3. The structure makes PCBs non-polar and insoluble in water, leading to bioaccumulation in fats.
4. The stability of these rings gives PCBs their high dielectric constant, making them suitable for electrical uses.
5. The environmental persistence of PCBs is linked to their chemical structure and chlorine substitutions.





 English
English 10 Questions
10 Questions 12 Mins
12 Mins