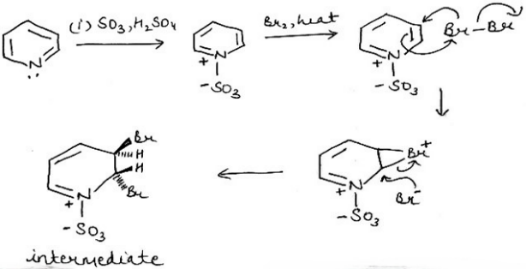
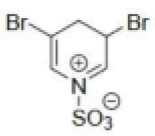
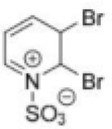
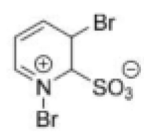
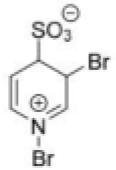
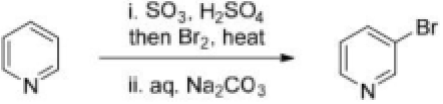Correct option is B
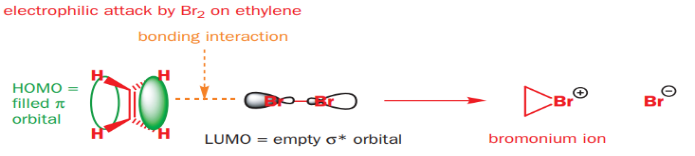
Lewis acids easily add to the nitrogen atom of pyridine, forming pyridinium salts. Evidently, neither the alkene nor bromine is charged, but Br2 has a low-energy empty orbital (the Br–Br σ*), and is therefore an electrophile. The alkene must be the nucleophile, and its HOMO is the C=C π bond. When it reacts with Br2 , the alkene’s filled π orbital (the HOMO) will interact with the bromine’s empty σ* orbital to give a product. The only way the π HOMO can interact in a bonding manner with the σ* LUMO is if the Br2 approaches end-on-and this is how the product forms. The symmetrical three-membered ring product is called a bromonium ion. Of course, the final product of the reaction isn’t the bromonium ion. The second step of the reaction follows on at once: the bromonium ion is an electrophile, and it reacts with the bromide ion lost from the bromine in the addition step. Overall, the molecule of bromine adds across the double bond of the alkene.
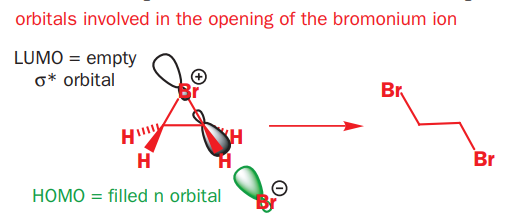
Attack of Br- on a bromonium ion is a normal SN2 substitution—the key orbitals involved are the HOMO of the bromide and the σ* of one of the two carbon–bromine bonds in the strained three-membered ring. As with all SN2 reactions, the nucleophile maintains maximal overlap with the σ* by approaching in line with the leaving group but from the opposite side, resulting in inversion at the carbon that is attacked.