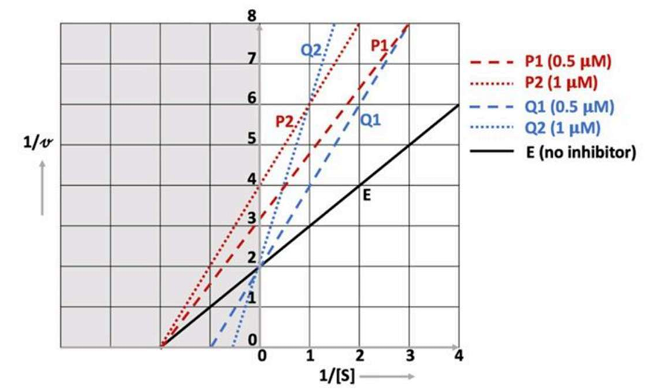
Correct option is D
Hemolysis of red blood cells (RBCs) occurs when they are placed in a hypotonic solution (i.e., a solution with a lower solute concentration than the cytoplasm of the RBCs). In such a condition, water moves into the RBCs by osmosis, causing them to swell and eventually burst.
- The normal physiological concentration of NaCl in human blood plasma is 0.9% NaCl (isotonic solution).
- A hypotonic solution has a lower NaCl concentration than 0.9%, leading to an influx of water into the RBCs.
- A hypertonic solution (higher than 0.9% NaCl) causes water to leave the RBCs, leading to shrinkage (crenation).
- When RBCs are placed in 0.5% NaCl, which is a hypotonic solution, water rushes into the cells due to osmosis, causing them to swell and eventually lyse (burst), resulting in hemolysis.
Thus, hemolysis occurs in 0.5% NaCl solution.
Information Booster
- Isotonic Solution (0.9% NaCl): No net water movement occurs, and RBCs maintain their normal shape.
- Hypertonic Solution (>0.9% NaCl, e.g., 2.0% or 1.5%): Water moves out of RBCs, causing shrinkage (crenation).
- Hypotonic Solution (<0.9% NaCl, e.g., 0.5%): Water moves into RBCs, leading to swelling and bursting (hemolysis).
- Hemolysis Mechanism: The osmotic influx of water increases intracellular pressure, eventually rupturing the plasma membrane.
- Medical Implications: Hemolysis can cause serious problems like hemoglobin release into plasma, potentially leading to kidney damage.
- Physiological Importance: Maintaining proper osmolarity in IV fluids prevents damage to blood cells.