Correct option is B
The correct answer is (b) Uracil
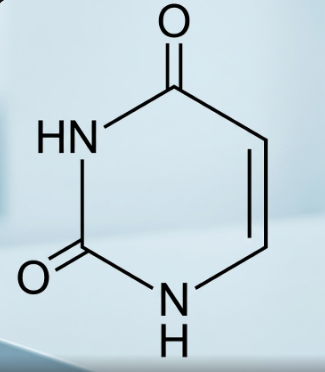
Explanation: • The image displays the chemical structure of
Uracil, which is one of the four nucleobases in the nucleic acid of
RNA (Ribonucleic Acid). • It is a
pyrimidine derivative, characterized by a single six-membered heterocyclic ring containing two nitrogen atoms at the 1 and 3 positions. • Chemically, it is known as
2,4-dioxopyrimidine because it features two
carbonyl (oxo) groups at the
C-2 and C-4 positions. • In the RNA structure,
Uracil forms two hydrogen bonds with Adenine during the process of base pairing. • It is typically absent in
DNA, where its functional role is replaced by
Thymine.
Information Booster: •
Thymine is essentially a
"methylated" version of Uracil; the presence of the methyl group in DNA helps the cell distinguish between natural bases and damaged bases (like deaminated cytosine). • In laboratory settings,
Uracil is used as a biochemical marker to help scientists track the synthesis and degradation of RNA in living organisms.
Additional Knowledge:
Adenine (Option a) • Unlike the single-ring pyrimidines,
Adenine is a
purine base with a
double-ring structure (a fused pyrimidine-imidazole ring system). • It is found in both
DNA and RNA and is a key component of
ATP (Adenosine Triphosphate).
Thymine (Option c) • Its structure is almost identical to
Uracil but contains an additional
methyl group (CH3) at the
C-5 position. • For this reason, it is chemically referred to as
5-methyluracil.
Cytosine (Option d) • This is also a
pyrimidine base, but instead of two carbonyl groups, it has an
amine group (NH2) at the
C-4 position. • It pairs with
Guanine through
three hydrogen bonds in both
DNA and RNA.





 English
English 25 Questions
25 Questions 20 Mins
20 Mins

