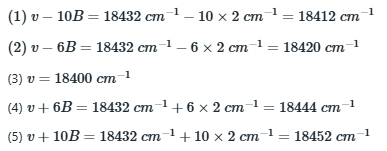Correct option is B
Quantum Theory of Raman Scattering. In contrast with other branches of spectroscopy, Raman spectroscopy deals with the scattering of light and not with its absorption. Consider a photon of frequency v falling on a molecule. If the collision is elastic, then the scattered photon will have the same energy as the incident photon. If, however, the collision is inelastic, the scattered photon will have either a higher or a lower energy than the incident photon. If we further assume that the total kinetic energy of the photon and the molecule remains unchanged before and after the collision, then, from the law of conservation of energy.
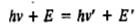
where hv is the energy of the incident photon and hv' is the energy of the scattered photon after collision; E is the energy of the molecule (rotational, vibrational and electronic) before collision and E is the molecular energy after collision.

Case I is referred to as Rayleigh scattering and cases II(a) and II(b) as Raman scattering. Thus, in the Rayleigh scattering, the scattered photon has the same frequency (or energy) as the incident photon while in the Raman scattering, when the photon collides with the molecule, the energy is either transferred to, or taken away from, the molecule. The Rayleigh scattering and the Raman scattering are shown in the figure
When the molecule, excited to the higher unstable vibrational state, returns to the original vibrational state, we get Rayleigh scattering. If it returns to a different vibrational state, this gives rise to Raman scattering (Stokes lines). When the molecule, initially in the first excited
vibrational state, is promoted to a higher unstable vibrational state and returns to the ground state, this again gives rise to Raman scattering (anti-Stokes lines). Thus, the Raman spectrum of a molecule consists of Stokes lines and anti-Stokes lines, situated symmetrically about the Rayleigh line.
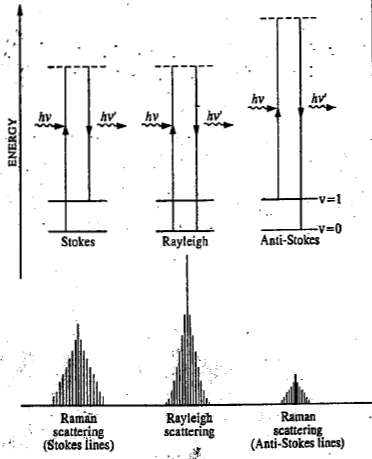
The Rayleigh line is far more intense than the Stokes lines which, in turn, have greater intensity than the anti-Stokes lines. The anti-Stokes lines are very difficult to observe in the conventional Raman spectroscopy because they correspond to the return of a molecule from the
unstable excited vibrational state to the ground state and initially there are very few molecules in the excited vibrational state.
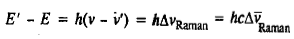


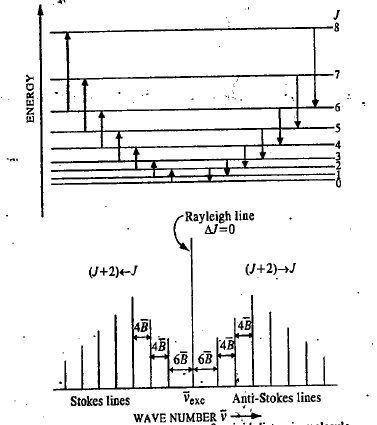
Rotational Raman Spectrum of a Diatomic Molecule
The selection rule for pure rotational Raman spectrum of a diatomic molecule is
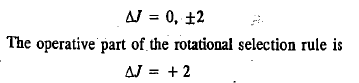
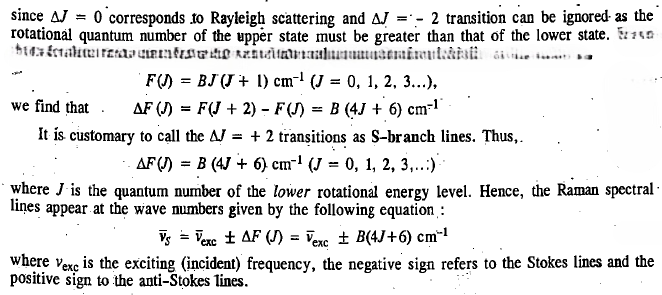
The first Stokes line (or the first anti-Stokes line) appears at a distance of 6B from the exciting Rayleigh line.