Correct option is B
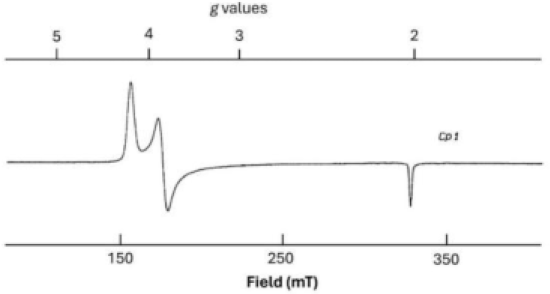
Electron Paramagnetic Resonance (EPR) spectroscopy is used to study systems with unpaired electrons. The g-value in EPR indicates the magnetic moment of the electron in a specific environment. In this question, the EPR spectrum corresponds to the FeMo cofactor in nitrogenase, which is responsible for nitrogen fixation in biological systems.
g-Values: The g-values shown in the spectrum indicate the interaction of the unpaired electron with the magnetic field. A typical g-value for a free electron is approximately 2.0023. Any deviation from this value indicates interactions with the surrounding atoms or ligands.
Spin State: The spin state of a system can be determined based on the number of unpaired electrons. In this case, the spin system corresponds to the overall spin (S).
Multiplicity: The number of signals or hyperfine splitting observed in the EPR spectrum gives insight into the spin multiplicity, which is related to the overall spin (S) by the equation (2S + 1).
The g-values (5, 4, 2) in the spectrum indicate multiple transitions associated with the FeMo cofactor's unpaired electrons. These g-values are typical for a system with multiple unpaired electrons and significant magnetic anisotropy.
The hyperfine splitting and g-values suggest that the system has an overall spin (S = 3/2). This corresponds to a quartet state, indicating three unpaired electrons in the system.
The spin multiplicity is calculated using the formula (2S + 1). For (S = 3/2), the multiplicity is 4, which aligns with the pattern observed in the EPR spectrum.