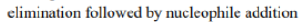Correct option is C
Indomethacin and its tetrazole analogue contain pyrrole rings with benzene rings fused to the side. Such bicyclic heterocyclic structures are called indoles. Indole itself has a benzene ring and a pyrrole ring sharing one double bond, or, if you prefer to look at it this way, it is an aromatic system with 10 electrons—eight from four double bonds and the lone pair from the nitrogen atom.
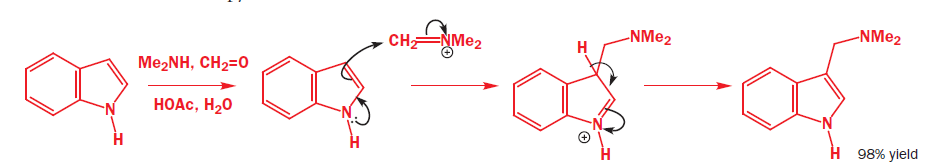
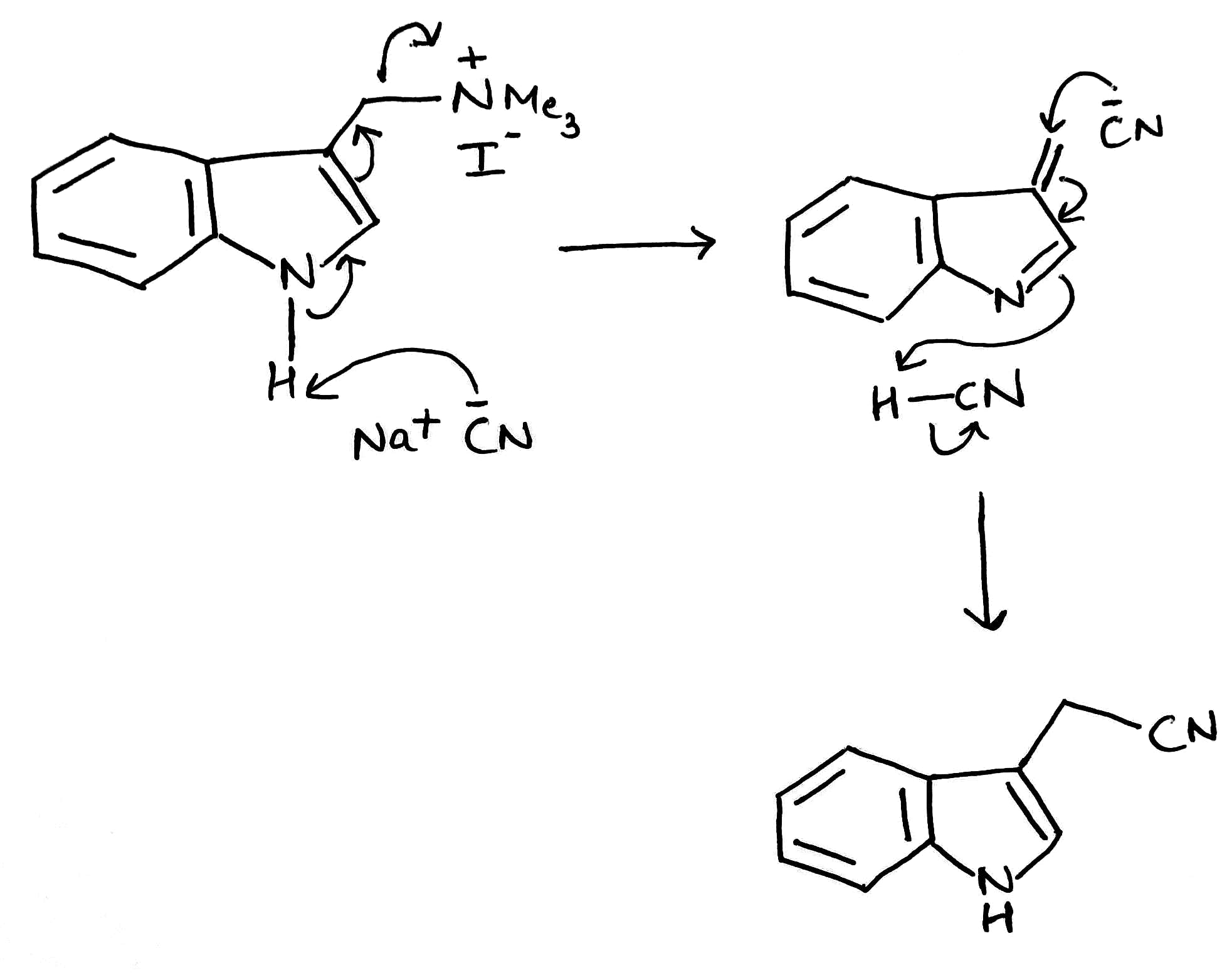
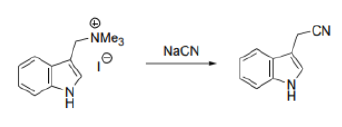
The indole nitrogen will expel the Me3N+group when NaCN is around as a base and nucleophile. The reaction is slow and the yield not wonderful but it is amazing that it happens at all. In the first step, CN- will act as a base and abstract the H+ .In the second step, CN- will act as a nucleophile and subsequently add to the double bond.