Correct option is D
A Boltzmann distribution is a probability distribution or probability measure that gives the probability that a system will be in a certain state as a function of that state's energy and the temperature of the system. The distribution is expressed in the form:
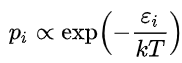
where pi is the probability of the system being in state i, exp is the exponential function, εi is the energy of that state, and a constant kT of the distribution is the product of the Boltzmann constant k and thermodynamic temperature T. The symbol ∝ denotes proportionality.
Two or more different states of a quantum mechanical system are said to be degenerate if they give the same value of energy upon measurement. The number of different states corresponding to a particular energy level is known as the degree of degeneracy (or simply the degeneracy) of the level.
Solution
The energy level having zero energy has two particles. The energy level having ε energy has two particles. Finally, the energy level having 2ε energy has two particles. Thus, total energy= 2×0+2×ε+2×2ε=6ε.








