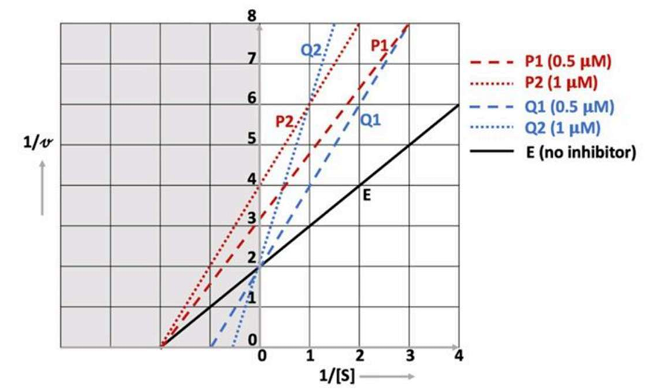
Correct option is A
Step 1: Understand the polysaccharide
So, 1 gram of this polysaccharide has:
- 1 gram of a polysaccharide made of 1000 glucose units means the polysaccharide is a single molecule with 1000 glucose monomers.
- The molecular weight of one glucose unit (C₆H₁₂O₆) is approximately 180 g/mol (6×12 for carbon + 12×1 for hydrogen + 6×16 for oxygen).
- However, in a polysaccharide, glucose units are linked by glycosidic bonds, and each bond formation releases a water molecule (H₂O, 18 g/mol). So, for 1000 glucose units, there are 999 glycosidic bonds formed, meaning 999 water molecules are lost.
- Molecular weight of 1000 glucose units without water loss:
1000×180=180,000g/mol1000 \times 180 = 180,000 \, \text{g/mol} 1000× 180=180,000g/mol. - Water loss from 999 bonds:
999×18=17,982g/mol999 \times 18 = 17,982 \, \text{g/mol} 999×18=17,982g/mol. - Molecular weight of the polysaccharide:
180,000−17,982=162,018g/mol180,000 - 17,982 = 162,018 \, \text{g/mol} 180,000−17,982=162,018g/mol.
Step 2: Osmolarity contribution of the polysaccharide
Osmolarity depends on the number of particles in solution. Since the polysaccharide is one molecule, 1 gram of it contributes:
6.17×10−6moles of particles6.17 \times 10^{-6} \, \text{moles of particles} 6.17×10-6 moles of particles.
Step 3: Osmolarity contribution of glucose
Glucose (C₆H₁₂O₆) has a molecular weight of 180 g/mol and does not dissociate in solution, so 1 mole of glucose contributes 1 mole of particles.
We need to find the mass of glucose that produces the same number of moles (and thus the same osmolarity) as the polysaccharide, i.e., 6.17×10−6moles 6.17 \times 10^{-6} \, \text{moles} 6.17×10-6
moles.
- Mass of glucose = moles × molecular weight: Mass=6.17×10-6
×180 ≈ 0.00111g=1.11mg. - The closest value to 1.11 mg is 1 mg.
- The correct option is 1. 1 mg glucose.