Correct option is C
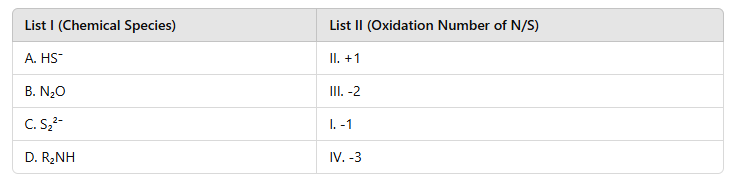
1. A. HS⁻ (Hydrosulfide ion) → II. +1: In HS⁻, hydrogen has an oxidation number of +1 because sulfur carries the -2 charge, balancing the ion's overall charge of -1.
2. B. N₂O (Nitrous oxide) → III. -2: The oxidation number of nitrogen (N) in N₂O is -2 when distributed between two nitrogen atoms, balancing the overall molecule.
3. C. S₂²⁻ (Disulfide ion) → I. -1: In the disulfide ion, each sulfur atom has an oxidation number of -1, sharing the overall -2 charge of the ion.
4. D. R₂NH (Secondary amine) → IV. -3: Nitrogen in amines generally has an oxidation state of -3 due to its bonding with hydrogen and carbon atoms.
Key Explanation: · HS⁻: Sulfur balances the hydrogen oxidation number of +1, making its state +1 in this molecule.
· N₂O: Nitrogen in N₂O is shared between two atoms, leading to an average oxidation state of -2 for the nitrogen atoms.
· S₂²⁻: Sulfur shares its oxidation state equally, with a charge of -1 per atom in the ion.
· R₂NH: The nitrogen atom typically has an oxidation state of -3 due to its bonding with alkyl or hydrogen groups.
Information Booster: 1. Oxidation Number Principles:
· Group 1 elements: Always +1.
· Hydrogen: +1 with nonmetals, -1 with metals.
· Oxygen: Typically -2 except in peroxides.
2. Special Cases in Oxidation:
· Disulfide ions (S₂²⁻) distribute charges between sulfur atoms equally.
· Nitrous oxide (N₂O) has shared oxidation states among nitrogen atoms.
3. Applications of Oxidation Numbers:
· Balancing chemical equations.
· Identifying oxidizing and reducing agents in reactions.





 English
English 10 Questions
10 Questions 12 Mins
12 Mins


