Correct option is C
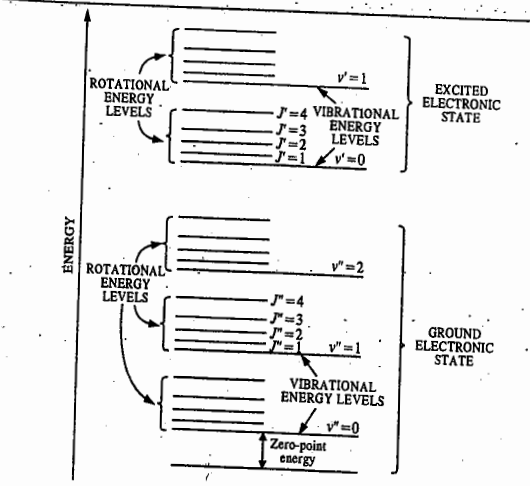
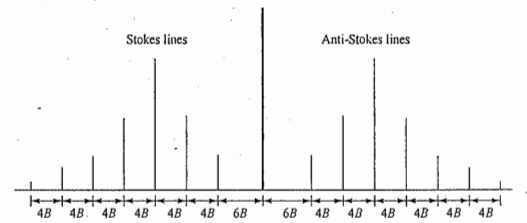
Raman spectroscopy is a spectroscopic technique typically used to determine vibrational modes of molecules, although rotational and other low-frequency modes of systems may also be observed. When the molecule, excited to the higher unstable vibrational state, returns to the original vibrational state, we get Rayleigh scattering. If it returns to a different vibrational state, this gives rise to Raman scattering (Stokes lines). When the molecule, initially in the first excited vibrational state, is promoted to a higher unstable vibrational state and returns to the ground state, this again gives rise to Raman scattering (anti-Stokes lines). Thus, the Raman spectrum of a molecule consists of Stokes lines and anti-Stokes lines, situated symmetrically about the Rayleigh line.
The selection rules for the pure rotational Raman spectra of diatomic molecules are ΔJ= 0 and ΔJ= ±2. The selection rule ΔJ= 0 gives rise to the Rayleigh scattering only. The selection rule ΔJ= ±2 gives rise to the Raman lines.