Correct option is B
Ans.(b)
For spontaneous reaction,
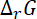
must be less than zero
So,
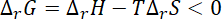
or,
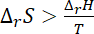
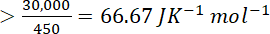
For reaction to be spontaneous the value of
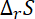
must be greater than 66.67 J that is 70 JK–1 mol–1
 for which the same reaction will be spontaneous at the same temperature is
for which the same reaction will be spontaneous at the same temperature is
Ans.(b)
For spontaneous reaction,
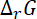
must be less than zero
So,
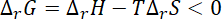
or,
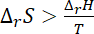
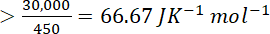
For reaction to be spontaneous the value of
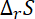
must be greater than 66.67 J that is 70 JK–1 mol–1
 English
English 200 Questions
200 Questions 200 Mins
200 Mins English
English English
English 200 Questions
200 Questions 200 Mins
200 Mins English
EnglishA gas stove flame burns blue because of:
Which is the chemical formula of salt?




A gas stove flame burns blue because of:
Suggested Test Series




Suggested Test Series



