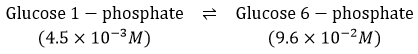Correct option is B
To calculate the equilibrium constant (Keq) and standard free energy change (ΔG°') for the given reaction, we can use the following formulas:
1. Equilibrium constant (Keq):
We can calculate the equilibrium constant using the formula:
Given:
[Glucose6−phosphate]=9.6×10−2M[Glucose 6-phosphate] = 9.6 \times 10^{-2} \, M[Glucose6−phosphate]=9.6×10-2 M
[Glucose1−phosphate]=4.5×10−3M[Glucose 1-phosphate] = 4.5 \times 10^{-3} \, M[Glucose1−phosphate]=4.5×10-3 M
2. Standard Free Energy Change (ΔG°'):
The standard free energy change (ΔG°') is related to the equilibrium constant (Keq) by the following equation:
Where:
R is the gas constant = 8.314 J/(mol·K)
T is the temperature in Kelvin. Since the problem gives the temperature as 25°C, we need to convert it to Kelvin:
T=25+273.15=298.15KT = 25 + 273.15 = 298.15 \, KT=25+273.15=298.15K =−8.314×298.15×ln(21.33)-8.314 \times 298.15 \times \ln(21.33)−8.314×298.15×ln(21.33)
where,
ln(21.33)≈3.063\ln(21.33) \approx 3.063ln(21.33)≈3.063
Therefore, the result is:
= -7595.2 J/Molar ,= -7.6 KJ/M
Final Answer:
The calculated values forKeqK_{eq}Keq and G∘\Delta G^\circΔG∘ are:
Keq =21.33
= -7.6 KJ/M