Correct option is B
Ans.(b)
Assuming dilute solution
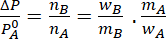
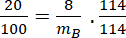
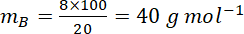
If 8g of a non-electrolyte solute is dissolved in 114g of n-octane to reduce its vapour pressure to 80%, the molar mass (in g mol⁻¹) of the solute is [Given that molar mass of n-octane is 114g mol⁻¹]
Ans.(b)
Assuming dilute solution
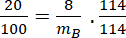
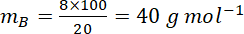
 English
English 200 Questions
200 Questions 200 Mins
200 Mins English
English English
English 200 Questions
200 Questions 200 Mins
200 Mins English
EnglishA gas stove flame burns blue because of:
Which is the chemical formula of salt?





Suggested Test Series




Suggested Test Series



