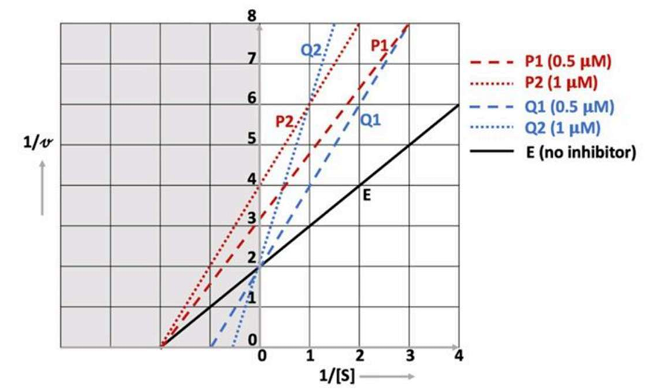
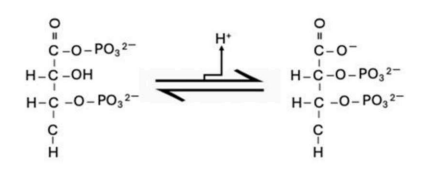
Correct option is B
Step 1: Calculate moles of enzyme
Mass of enzyme = 10 µg = 10 × 10⁻⁶ g
Molar mass of enzyme = 30,000 g/mol
Moles of enzyme = (10 × 10⁻⁶) / 30,000
Moles of enzyme = 3.33 × 10⁻¹⁰ mol
Step 2: Calculate moles of CO₂ converted
Mass of CO₂ = 0.30 g
Molar mass of CO₂ = 44 g/mol
Moles of CO₂ = 0.30 / 44
Moles of CO₂ = 6.82 × 10⁻³ mol
Step 3: Turnover number (kcat)
kcat = (moles of substrate converted per minute) / (moles of enzyme)
kcat = (6.82 × 10⁻³) / (3.33 × 10⁻¹⁰)
kcat ≈ 2.05 × 10⁷ min⁻¹
Final Answer:
Turnover number, kcat = 2.05 × 10⁷ min⁻¹