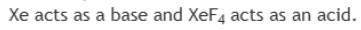Correct option is D
Xenon fluorides are strong oxidizing agents and form complexes with,
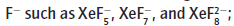
they are used in the preparation of compounds containing Xe-O and Xe-N bonds.
The reactions of the xenon fluorides are similar to those of the high oxidation state interhalogens, and redox and metathesis reactions dominate.
The reaction of XeF4 with the Lewis base F- in acetonitrile (cyanomethane,
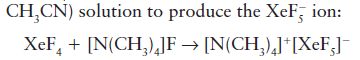
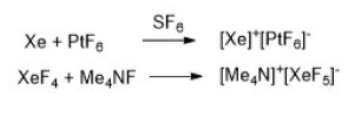
In the above reaction, XeF4 acts as an acid. A Lewis acid is a chemical species that contains an empty orbital which is capable of accepting an electron pair from a Lewis base to form a Lewis adduct. A Lewis base, then, is any species that has a filled orbital containing an electron pair which is not involved in bonding but may form a dative bond with a Lewis acid to form a Lewis adduct.
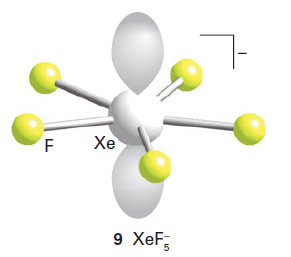
The XeF5- ion is pentagonal planar, and in the VSEPR model the two electron pairs on Xe occupy axial positions on opposite sides of the plane.
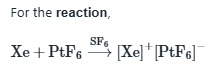
Xe donates an electron to form a monopositive ion (Xe+) and PtF6 accepts an electron to form a mononegative ion [PtF6]-.