Correct option is B
The correct option is (b)
Explanation:
1. Basic Thermodynamics Relation:
The standard Gibbs free energy change is related to the equilibrium constant by the equation:
where,
R is the universal gas constant,
T is the absolute temperature in Kelvin,
ln is the natural logarithm.
2. Given Condition:
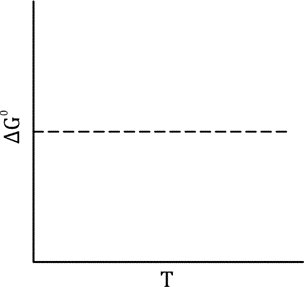
From the graph , does not change with temperature T. This implies:
3. Finding
Substitute into the thermodynamic relation
Rearranging :
4. Exponentiating both sides:
5. Interpreting the result:
This shows that depends exponentially on when is constant
So the correct relation is: