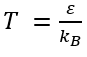Correct option is D
A partition function describes the statistical properties of a system in thermodynamic equilibrium. Partition functions are functions of the thermodynamic state variables, such as the temperature and volume. Most of the aggregate thermodynamic variables of the system, such as the total energy, free energy, entropy, and pressure, can be expressed in terms of the partition function or its derivatives. The partition function is dimensionless.
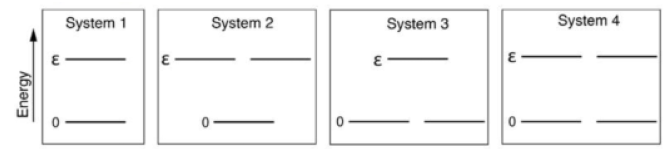
In systems with multiple quantum states s sharing the same energy Es , it is said that the energy levels of the system are degenerate. Two or more different states of a quantum mechanical system are said to be degenerate if they give the same value of energy upon measurement. In the case of degenerate energy levels, the partition function can be written in terms of the contribution from energy levels (indexed by j) as follows:
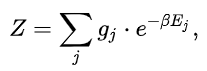
where
· j is the index for the microstates of the system (a microstate is a specific configuration of a system that describes the precise positions and momenta of all the individual particles or components that make up the system);
· e is Euler's number
· β is the thermodynamic beta, defined as 1/kBT where kB is the Boltzmann constant;
· Ej is the total energy of the system in the respective microstate.
· gj is the degeneracy factor, or number of quantum states s that have the same energy level defined by Ej = Es .
The Helmholtz free energy (F) is a thermodynamic potential that measures the useful work obtainable from a closed thermodynamic system at a constant temperature (isothermal).

where
F is the Helmholtz free energy (sometimes also called A, particularly in the field of chemistry)
T is the absolute temperature (kelvins) of the surroundings, modelled as a heat bath