Correct option is D
Explanation-
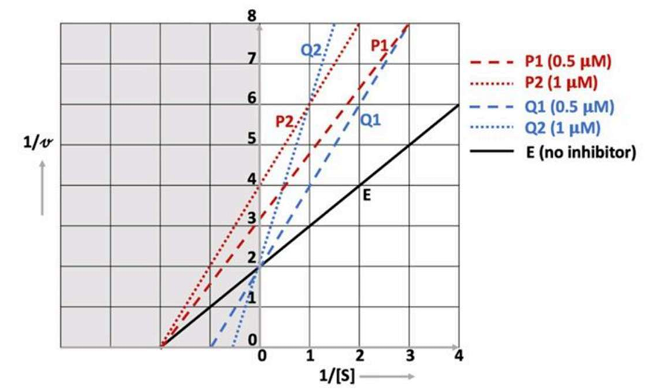
The pI is the pH at which the amino acid carries no net electric charge (zwitterion). For amino acids with ionizable side chains (like lysine), we use the two pKa values surrounding the neutral (zwitterionic) form.
Lysine Ionization States
Lysine has 3 ionizable groups, so there are 4 ionization states:
Fully protonated form (Net charge: +2)
Zwitterion (Net charge: +1) — this is where pI is between the two pKa values where the molecule goes from +2 → 0.
Neutral form (Net charge: 0) – occurs between the two highest pKa values, pKa2 (9.06) and pKa3 (10.54).
Net -1 form
pI Formula for Lysine:
Since lysine is a basic amino acid, we calculate the pI using the two highest pKa values:
So, the correct answer is option d : 9.8