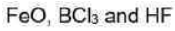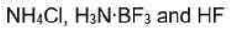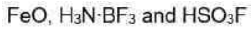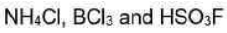Correct option is D
An acid is a molecule or ion capable of either donating a proton (i.e. hydrogen ion, H+), known as a Brønsted–Lowry acid, or forming a covalent bond with an electron pair, known as a Lewis acid. In the special case of aqueous solutions, proton donors form the hydronium ion H3O+ and are known as Arrhenius acids.
(i) Ammonium chloride is an inorganic chemical compound with the chemical formula NH4Cl, also written as [NH4] Cl. It is an ammonium salt of hydrogen chloride. It consists of ammonium cations [NH4]+ and chloride anions Cl-.It is a white crystalline salt that is highly soluble in water. Solutions of ammonium chloride are mildly acidic.
Ammonium chloride is prepared commercially by combining ammonia (NH3) with either hydrogen chloride (gas) or hydrochloric acid (water solution):
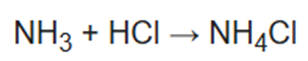
Iron (II) oxide or ferrous oxide is the inorganic compound with the formula FeO. It is insoluble in alkali, alcohol. FeO dissolves in acid.
(ii) In coordination chemistry, a coordinate covalent bond, also known as a dative bond, dipolar bond, or coordinate bond is a kind of two-center, two-electron covalent bond in which the two electrons derive from the same atom. The bonding of metal ions to ligands involves this kind of interaction. This type of interaction is central to Lewis acid–base theory.
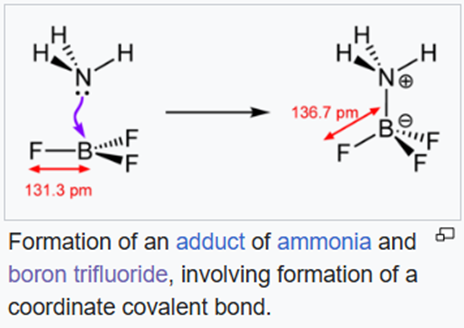
All three lighter boron trihalides, BX3 (X = F, Cl, Br) form stable adducts with common Lewis bases. Their relative Lewis acidities can be evaluated in terms of the relative exothermicities of the adduct-forming reaction. Such measurements have revealed the following sequence for the Lewis acidity:
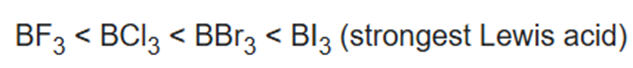
This trend is commonly attributed to the degree of π-bonding in the planar boron trihalide that would be lost upon pyramidalization of the BX3 molecule which follows this trend:

The criteria for evaluating the relative strength of π-bonding are not clear, however. One suggestion is that the F atom is small compared to the larger Cl and Br atoms. As a consequence, the bond length between boron and the halogen increases while going from fluorine to iodine hence spatial overlap between the orbitals becomes more difficult. The lone pair electron in pz of F is readily and easily donated and overlapped to empty pz orbital of boron. As a result, the pi donation of F is greater than that of Cl or Br.
In an alternative explanation, the low Lewis acidity for BF3 is attributed to the relative weakness of the bond in the adducts F3B−L.
(iii) Fluorosulfuric acid is the inorganic compound with the chemical formula HSO3F. It is one of the strongest acids commercially available. It is a tetrahedral molecule and is closely related to sulfuric acid, H2SO4, substituting a fluorine atom for one of the hydroxyl groups.
HSO3F is one of the strongest known simple Brønsted acids. The combination of HSO3F and the Lewis acid antimony pentafluoride produces "Magic acid", which is a far stronger protonating agent. These acids are categorized as "superacids", acids stronger than 100% sulfuric acid.
Reflecting its strong acidity, HSO3F dissolves almost all organic compounds that are even weak proton acceptors.
Hydrofluoric acid is a solution of hydrogen fluoride (HF) in water. Solutions of HF are colorless, acidic and highly corrosive.
In dilute aqueous solution hydrogen fluoride behaves as a weak acid. Infrared spectroscopy has been used to show that, in solution, dissociation is accompanied by formation of the ion pair H3O+·F-.
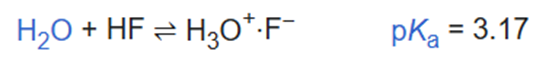
This ion pair has been characterized in the crystalline state at very low temperature. Further association has been characterized both in solution and in the solid state.
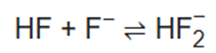
Unlike other hydrohalic acids, such as hydrochloric acid, hydrogen fluoride is only a weak acid in dilute aqueous solution. This is in part a result of the strength of the hydrogen–fluorine bond, but also of other factors such as the tendency of HF, H2O, and F- anions to form clusters. At high concentrations, HF molecules undergo homoassociation to form polyatomic ions (such as bifluoride, HF2- ) and protons, thus greatly increasing the acidity. This leads to protonation of very strong acids like hydrochloric, sulfuric, or nitric acids when using concentrated hydrofluoric acid solutions. Although hydrofluoric acid is regarded as a weak acid, it is very corrosive, even attacking glass when hydrated.