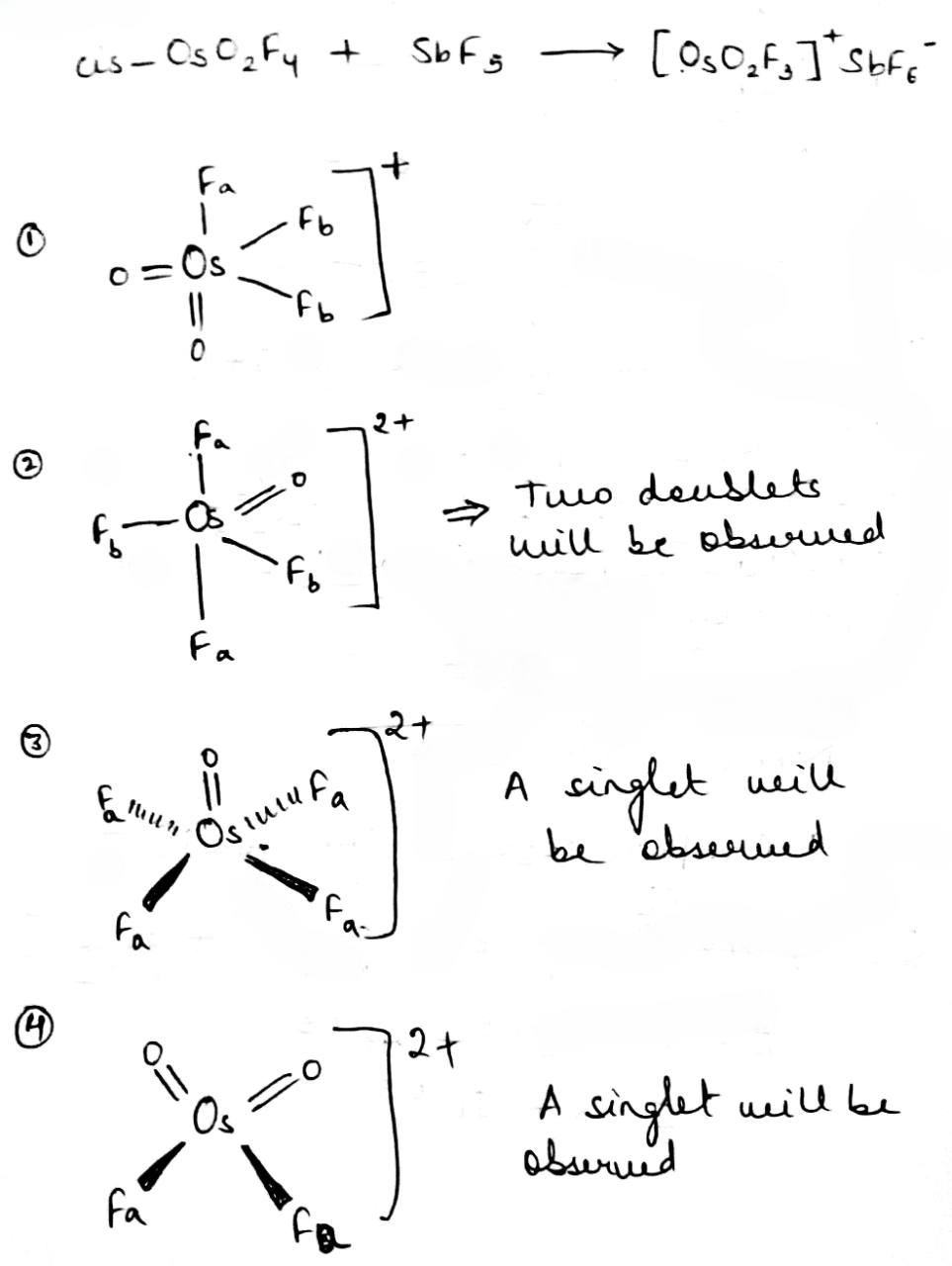
Correct option is A
Fluorine-19 nuclear magnetic resonance spectroscopy (fluorine NMR or 19F NMR) is an analytical technique used to detect and identify fluorine-containing compounds. Fluorine atoms can couple with each other.
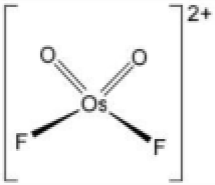
[OsO2F3]+ has a trigonal bipyramidal geometry. Osmium shares a plane with two fluorine atoms and an oxygen at 120° angles to each other in equatorial positions, and one fluorine atom above and one oxygen atom below the plane ( axial or apical positions). The 19F NMR spectrum helps in predicting the structure based on the chemical environment of the fluorine atoms. There are two kinds of fluorine atoms present in the Os species. Fa will couple with two Fb and will give a triplet. Two similar Fb will couple with one Fa and will give a doublet.