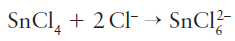Correct option is B
Any substance that acts as a proton donor should be classified as an acid, and any substance that acts as a proton acceptor should be classified as a base. Substances that act in this way are called ‘Brønsted acids’ and ‘Brønsted bases’, respectively:
A Brønsted acid is a proton donor. A Brønsted base is a proton acceptor.
Aluminosilicate refers to materials containing anionic Si-O-Al linkages. Commonly, the associate cations are sodium (Na+), potassium (K+) and protons (H+). Such materials occur as minerals, coal combustion products and as synthetic materials, often in the form of zeolites. Aluminosilicates are widely used in the field of catalysis due to their Brønsted acidic properties that enable the conversion of hydrocarbons.
A Lewis acid is a substance that acts as an electron pair acceptor. A Lewis base is a substance that acts as an electron pair donor. We denote a Lewis acid by A and a Lewis base by :B, often omitting any other lone pairs that may be present. The fundamental reaction of Lewis acids and bases is the formation of a complex (or adduct), A–B, in which A and :B bond together by sharing the electron pair supplied by the base.
Group 14 elements other than carbon exhibit hypervalence and act as Lewis acids by becoming five- or six-coordinate; tin(II) chloride is both a Lewis acid and a Lewis base.
Unlike carbon, a Si atom can expand its valence shell (or is simply large enough) to become hypervalent. For example, a five-coordinate trigonal bipyramidal structure is possible . A representative Lewis acid–base reaction is that of SiF4 with two F- ions:

The trend in acidity for SiX4, which follows the order SiI4 (3p-5p)< SiBr4(3p-4p) < SiCl4(3p-3p)< SiF4(3p-2p), correlates with the increase in the electron-withdrawing power of the halogen from I to F.
Tin(II) chloride is both a Lewis acid and a Lewis base. As an acid, SnCl2 combines with Cl- to form SnCl3- . This complex retains a lone pair, and it is sometimes more revealing to write its formula as :SnCl3-. It acts as a base to give metal–metal bonds, as in the complex
(CO)5Mn–SnCl3. Tin(IV) halides are Lewis acids. They react with halide ions to form SnX62-: